(3S,4S)-4-((R)-2-(Benzyloxy)tridecyl)-3-hexyloxetan-2-one
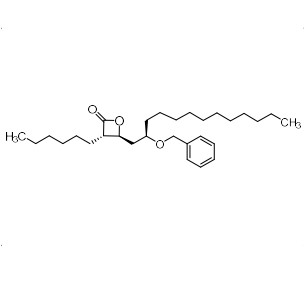
M.F. C₂₉H₄₈O₃
M.W. 444.70
CAT# AR-O08825
CAS# 114264-05-0

M.F. C₂₉H₄₈O₃
M.W. 444.70
CAT# AR-O08825
CAS# 114264-05-0

M.F. C₃₀H₅₃NO₃
M.W. 475.76
CAT# AR-O08826
CAS# NA
![Show details for N-Formyl-L-leucine(1S)-1-[(2S,3S)-2-hydroxy-3-[(R)-1-phenylethylcarbomoyl)]nonyl]dodecyl Ester Picture of N-Formyl-L-leucine(1S)-1-[(2S,3S)-2-hydroxy-3-[(R)-1-phenylethylcarbomoyl)]nonyl]dodecyl Ester](https://bc3c83a0874864a50753-7010fd7f2bdf81ab41ee28479491701e.ssl.cf2.rackcdn.com/media//catalog/AR-O00891.jpg?size=256)
M.F. C₃₇H₆₄N₂O₅
M.W. 616.91
CAT# AR-O00891
CAS# NA

M.F. C₂₂H₄₄O₄
M.W. 372.59
CAT# AR-O01457
CAS# 145682-69-5

M.F. C₂₉H₅₅NO₆
M.W. 513.75
CAT# AR-O00992
CAS# NA

M.F. C₂₂H₄₂O₃
M.W. 354.57
CAT# AR-O00993
CAS# NA

M.F. C₂₂H₄₂O₃
M.W. 354.57
CAT# AR-O00994
CAS# NA
![Show details for N-formyl-L-Leucine(S)-1-[(2S,3S)-3-octyl-4-oxo-oxetan-2-ylmethyl]-dodecyl ester Picture of N-formyl-L-Leucine(S)-1-[(2S,3S)-3-octyl-4-oxo-oxetan-2-ylmethyl]-dodecyl ester](https://bc3c83a0874864a50753-7010fd7f2bdf81ab41ee28479491701e.ssl.cf2.rackcdn.com/media//catalog/AR-O00996.jpg?size=256)
M.F. C₃₁H₅₇NO₅
M.W. 523.79
CAT# AR-O00996
CAS# NA
![Show details for N-formyl-L-valine(S)-1-[(2S,3S)-3-hexyl-4-oxo-oxetan-2-ylmethyl]-dodecyl ester Picture of N-formyl-L-valine(S)-1-[(2S,3S)-3-hexyl-4-oxo-oxetan-2-ylmethyl]-dodecyl ester](https://bc3c83a0874864a50753-7010fd7f2bdf81ab41ee28479491701e.ssl.cf2.rackcdn.com/media//catalog/AR-O00998.jpg?size=256)
M.F. C₂₈H₅₁NO₅
M.W. 481.71
CAT# AR-O00998
CAS# NA

M.F. C₂₂H₄₃O₄. Na
M.W. 371.59 22.99
CAT# AR-O01442
CAS# 130793-30-5 (free acid)
Can't find what you are looking for?
Inquire Now