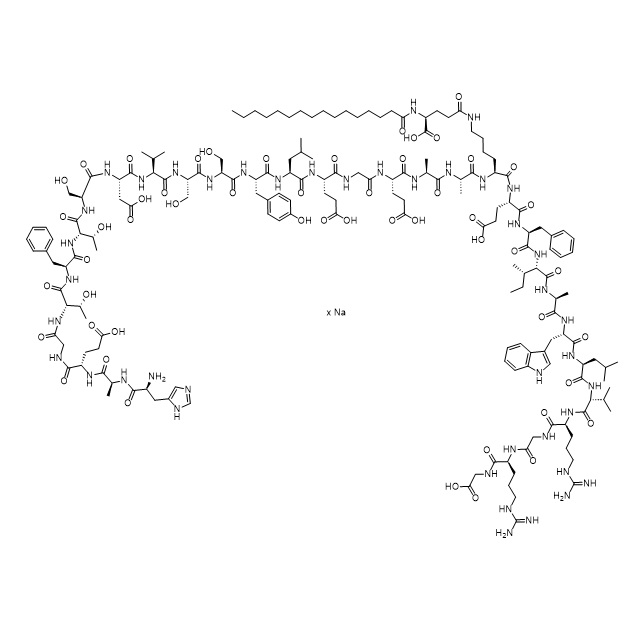
- SynonymsN5-((2S,5S,8S,11S,14S,17S)-17-((S)-2-((S)-2-((S)-5-amino-2-(2-((S)-2-((S)-2-((S)-2-((S)-2-amino-3-hydroxypropanamido)-3-(4-hydroxyphenyl)propanamido)-4-methylpentanamido)-4-carboxybutanamido)acetamido)-5-oxopentanamido)propanamido)propanamido)-11-benzyl-8-((S)-sec-butyl)-14-(2-carboxyethyl)-2-(((...
- Description
N5-((2S,5S,8S,11S,14S,17S)-17-((S)-2-((S)-2-((S)-5-amino-2-(2-((S)-2-((S)-2-((S)-2-((S)-2-amino-3-hydroxypropanamido)-3-(4-hydroxyphenyl)propanamido)-4-methylpentanamido)-4-carboxybutanamido)acetamido)-5-oxopentanamido)propanamido)propanamido)-11-benzyl-8-((S)-sec-butyl)-14-(2-carboxyethyl)-2-(((S)-1-(((S)-1-(((S)-1-((2-(((S)-1-((carboxymethyl)amino)-5-guanidino-1-oxopentan-2-yl)amino)-2-oxoethyl)amino)-5-guanidino-1-oxopentan-2-yl)amino)-3-methyl-1-oxobutan-2-yl)amino)-4-methyl-1-oxopentan-2-yl)carbamoyl)-1-(1H-indol-3-yl)-5-methyl-4,7,10,13,16-pentaoxo-3,6,9,12,15-pentaazahenicosan-21-yl)-N2-palmitoyl-L-glutamine
Liraglutide Hydrolysis 18-37 Impurity is a fully characterized chemical compound used as a reference standard of API Lumacaftor. The standard offered is compliant with regulatory guidelines. Liraglutide Hydrolysis 18-37 Impurity is used for analytical method development, method validation (AMV), and Quality Controlled (QC) applications during synthesis and formulation stages of drug development and serves as a reference standard for traceability against pharmacopeial standards (USP or EP). Axios Research products are intended for analytical purposes only and are not for human use. CAS - NA
Related products
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
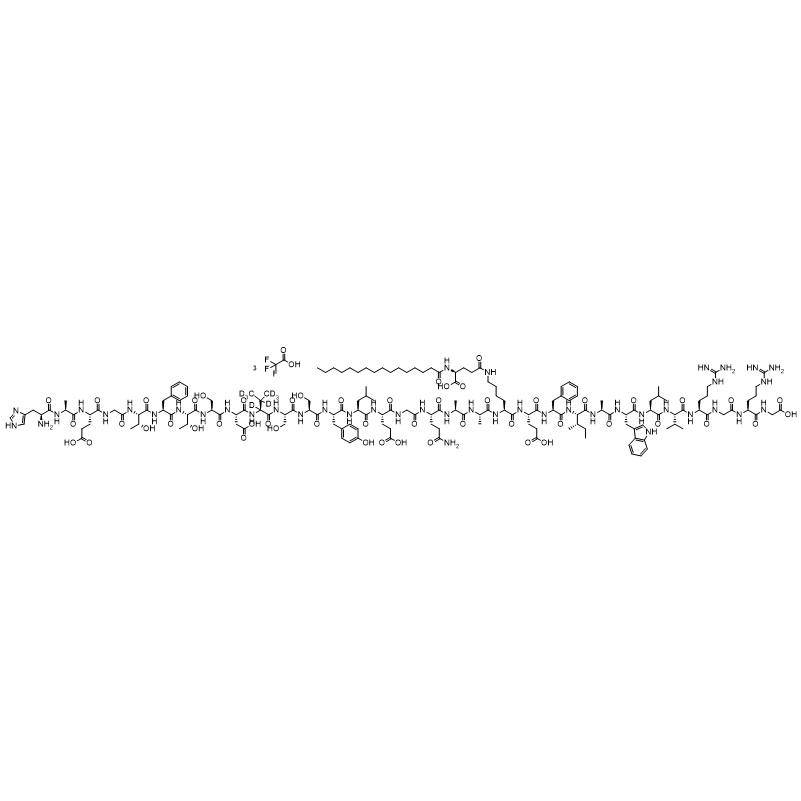
Liraglutide Impurity 2 TFA (Kyn25-Liraglutide TFA)

M.F.
M.W. 3755.25 4*114.02
CAT# AR-L07491
CAS# NA
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...
Liraglutide
More options:
Enter Qty (# of units), Enter Qty (mg), Special...













![Show details for Liraglutide Impurity 1 ([Trp(O)]25-Liraglutide) Picture of Liraglutide Impurity 1 ([Trp(O)]25-Liraglutide)](https://bc3c83a0874864a50753-7010fd7f2bdf81ab41ee28479491701e.ssl.cf2.rackcdn.com/media//catalog/AR-L07514.JPG?size=256)











![Show details for Liraglutide [7-31] Fragment Picture of Liraglutide [7-31] Fragment](https://bc3c83a0874864a50753-7010fd7f2bdf81ab41ee28479491701e.ssl.cf2.rackcdn.com/media//catalog/AR-L07505.JPG?size=256)
![Show details for Liraglutide [3-31] Fragment Picture of Liraglutide [3-31] Fragment](http://axios-research.com/media/0/default-image.png?size=256)