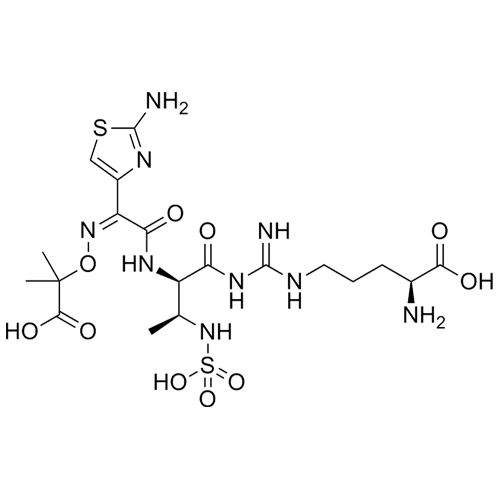
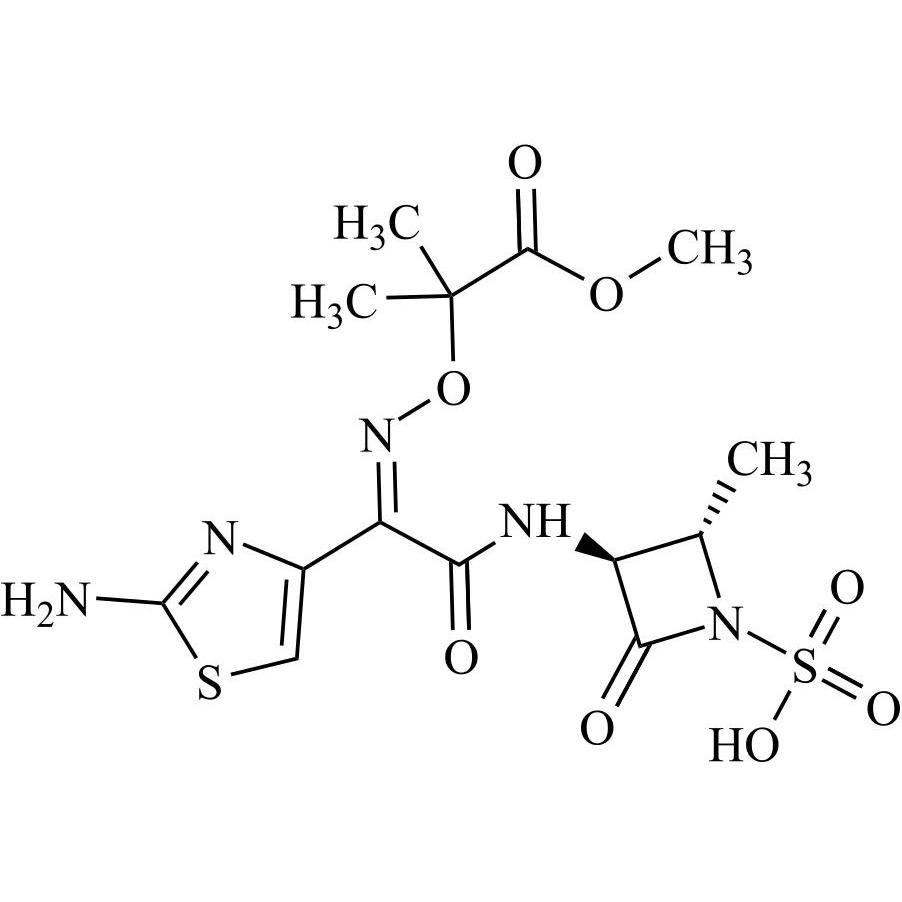
- Synonyms2-(((Z)-(1-(5-(((2R,3S)-2-((Z)-2-(2-aminothiazol-4-yl)-2-(((2-carboxypropan-2-yl)oxy)imino)acetamido)-3-(sulfoamino)butanoyl)oxy)thiophen-3-yl)-2-(((2S)-2-methyl-4-oxo-1-sulfoazetidin-3-yl)amino)-2-oxoethylidene)amino)oxy)-2-methylpropanoic acid
- Description
2-(((Z)-(1-(5-(((2R,3S)-2-((Z)-2-(2-aminothiazol-4-yl)-2-(((2-carboxypropan-2-yl)oxy)imino)acetamido)-3-(sulfoamino)butanoyl)oxy)thiophen-3-yl)-2-(((2S)-2-methyl-4-oxo-1-sulfoazetidin-3-yl)amino)-2-oxoethylidene)amino)oxy)-2-methylpropanoic acid
Aztreonam Impurity 4 is a fully characterized chemical compound used as a reference standard of API Aztreonam. The standard offered is compliant with regulatory guidelines. Aztreonam Impurity 4 is used for analytical method development, method validation (AMV), and Quality Controlled (QC) applications during synthesis and formulation stages of drug development and serves as a reference standard for traceability against pharmacopeial standards (USP or EP). Axios Research products are intended for analytical purposes only and are not for human use. CAS -
Related products
Aztreonam Impurity 1 Disodium Salt (Mixture of Diastereomers)
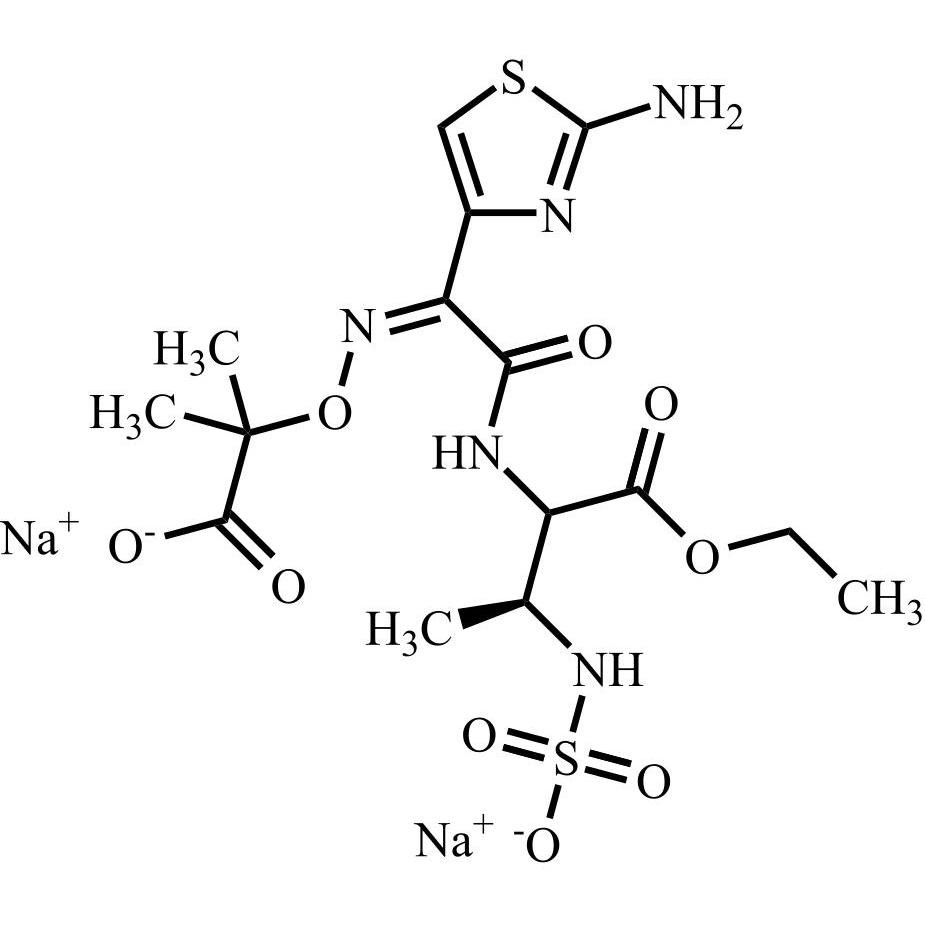
M.F.
M.W. 479.49 2*22.99
CAT# AR-A07996
CAS# NA
Aztreonam USP Related Compound B Monosodium Salt (Open-ring Desulfated Aztreonam Monosodium Salt)
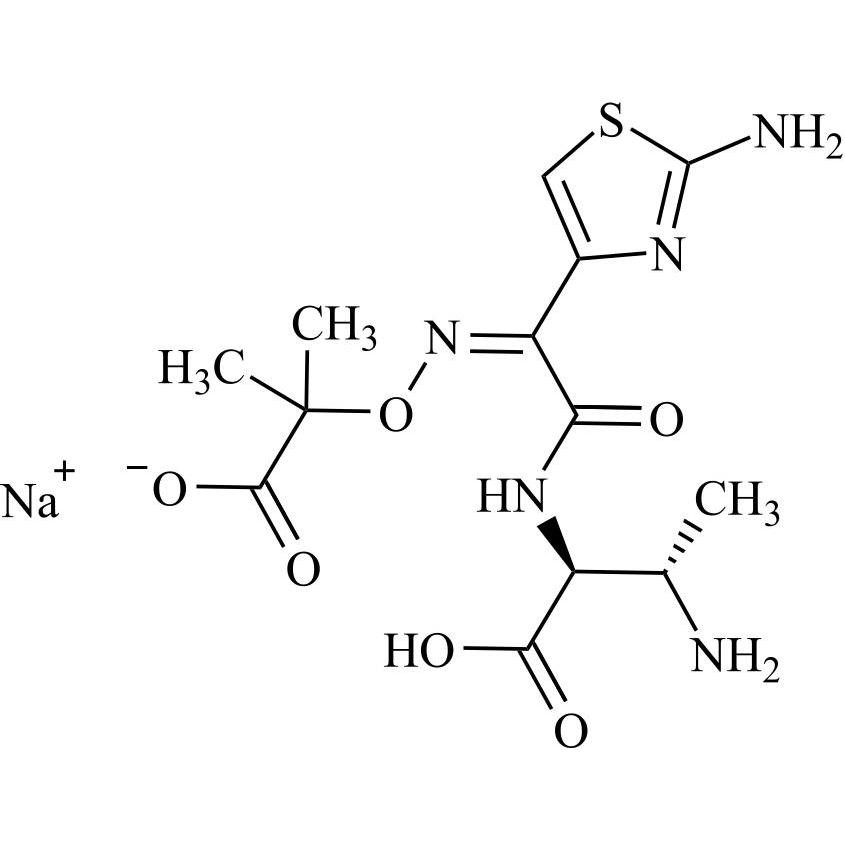
M.F.
M.W. 372.38 22.99
CAT# AR-A07993
CAS# NA
Aztreonam USP Related Compound D Sodium Salt (Desulfated Aztreonam Sodium Salt)
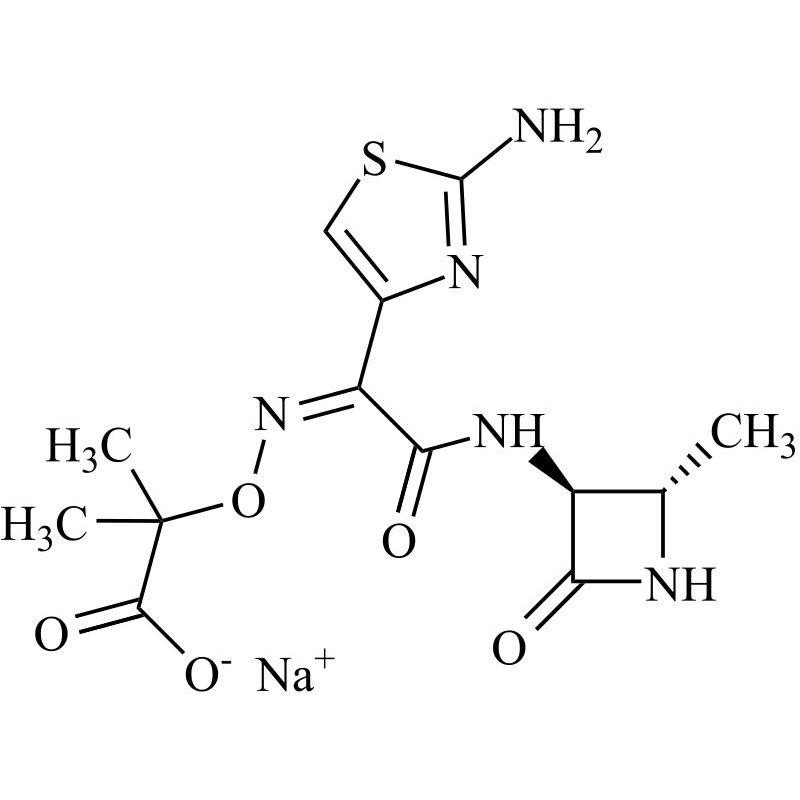
M.F.
M.W. 354.36 22.99
CAT# AR-A07994
CAS# NA