(R)-(3-methyl-1-(pyrazine-2-carboxamido)butyl)boronic acid
Bortezomib Impurity 7 is a fully characterized chemical compound used as a reference standard of API Bortezomib. The standard offered is compliant with regulatory guidelines. Bortezomib Impurity 7 is used for analytical method development, method validation (AMV), and Quality Controlled (QC) applications during synthesis and formulation stages of drug development and serves as a reference standard for traceability against pharmacopeial standards (USP or EP). Axios Research products are intended for analytical purposes only and are not for human use. CAS - N/A
Related products
Bortezomib Hydroperoxide Mixture. 1:1

M.F.
M.W. 372.43
CAT# AR-B01898
CAS# 886979-78-8 + 886979-81-3
(R)-(1-amino-3-methylbutyl)boronic acid hydrochloride
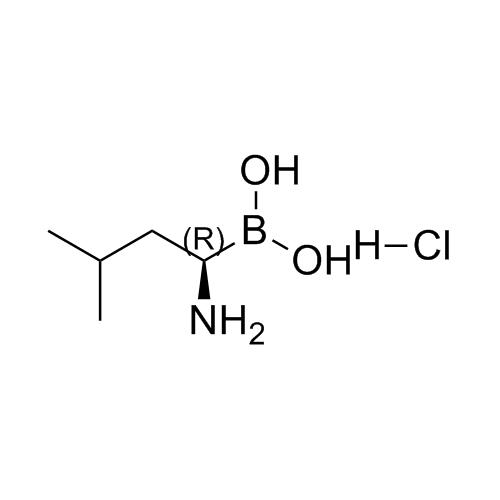
M.F.
M.W. 130.98 36.46
CAT# AR-B01899
CAS# 1158974-93-6
((R)-3-Hydroxy-3-methyl-1-((S)-3-phenyl-2-(pyrazine-2-carboxamido)propanamido)butyl)boronic acid
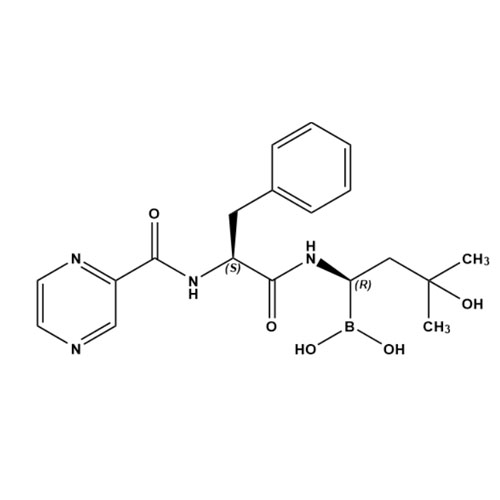
M.F.
M.W. 400.25
CAT# AR-B05560
CAS# NA
Bortezomib Impurity ((S)-3-Phenyl-2- (pyrazine-2-carbonylamino) propanoic Acid)

M.F.
M.W. 271.28
CAT# AR-B01842
CAS# 114457-94-2
Bortezomib Impurity (Mixture of (1S,2R)-Isomer and (1R,2R)-Isomer)
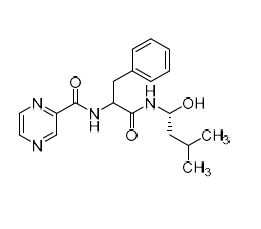
M.F.
M.W. 356.43
CAT# AR-B01869
CAS# 2747958-64-9
Bortezomib Mixture of Isomer (SS and RR, 1:1)
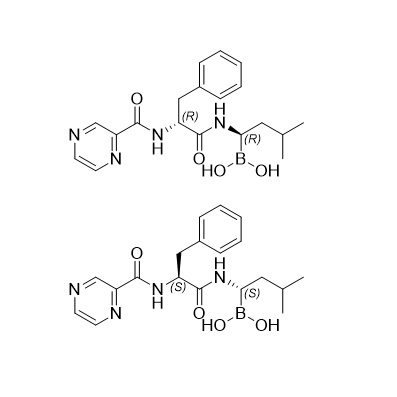
M.F.
M.W. 384.2
CAT# AR-B08224
CAS# 1132709-15-9 + 1132709-14-8




































































































