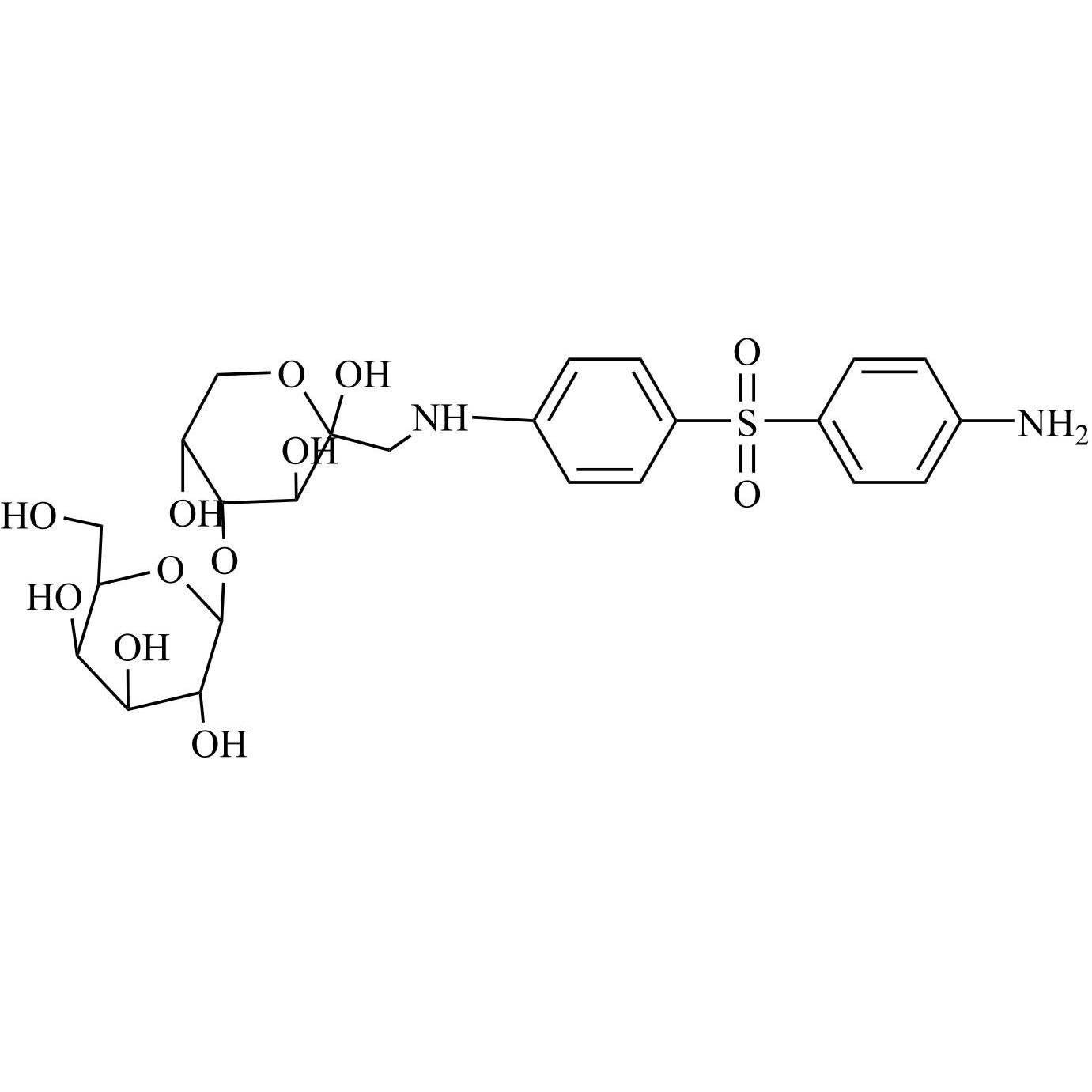
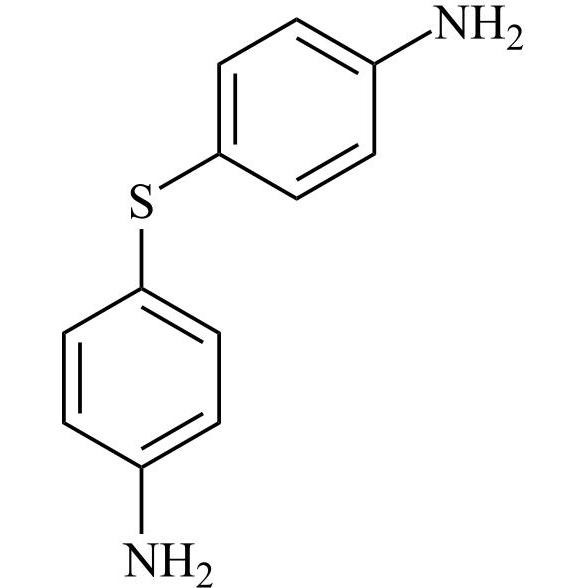
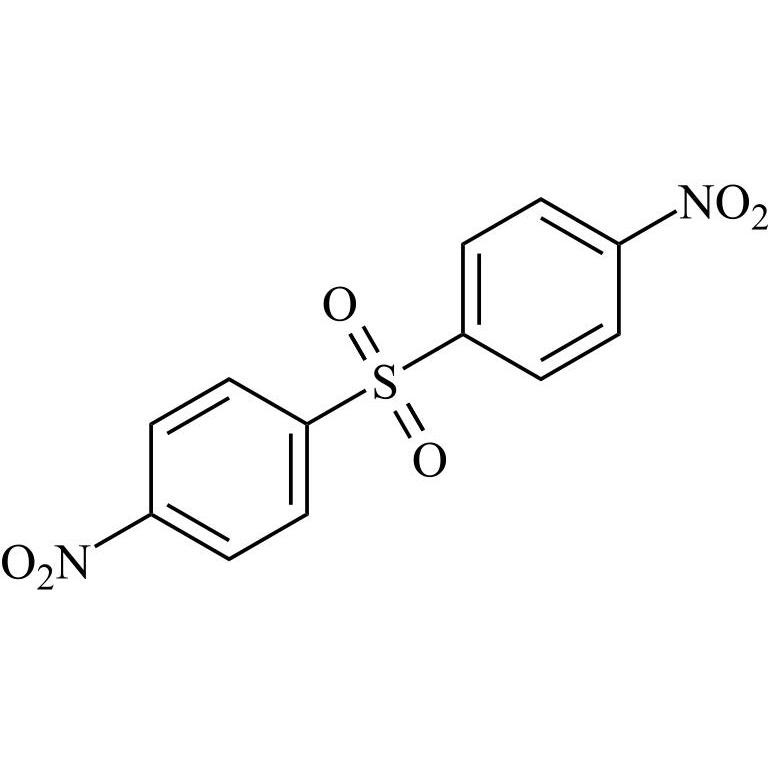
- Synonyms4-((4-chlorophenyl)sulfonyl)aniline;4-[(4-Chlorophenyl)sulfonyl]benzenamine; p-[(p-Chlorophenyl)sulfonyl]aniline; 4-(4-Chlorobenzenesulfonyl)phenylamine; 4-Amino-4'-chlorodiphenyl Sulfone; 4-Aminophenyl 4-chlorophenyl Sulfone; NSC 23812; NSC 51990; p-(p-Chlorophenylsulfonyl)aniline; p-Aminophenyl...
- Description
4-((4-chlorophenyl)sulfonyl)aniline;4-[(4-Chlorophenyl)sulfonyl]benzenamine; p-[(p-Chlorophenyl)sulfonyl]aniline; 4-(4-Chlorobenzenesulfonyl)phenylamine; 4-Amino-4'-chlorodiphenyl Sulfone; 4-Aminophenyl 4-chlorophenyl Sulfone; NSC 23812; NSC 51990; p-(p-Chlorophenylsulfonyl)aniline; p-Aminophenyl p-Chlorophenyl Sulfone;4-Amino-4'-chlorodiphenyl Sulfone
Dapsone EP Impurity E is a fully characterized chemical compound used as a reference standard of API Dapsone. The standard offered is compliant with regulatory guidelines. Dapsone EP Impurity E is used for analytical method development, method validation (AMV), and Quality Controlled (QC) applications during synthesis and formulation stages of drug development and serves as a reference standard for traceability against pharmacopeial standards (USP or EP). Axios Research products are intended for analytical purposes only and are not for human use. CAS - 7146-68-1