- Synonyms1,3-Diaza-2,4-cyclopentadiene; Ondansetron EP Impurity E; Clotrimazole EP Impurity D
- Description
1,3-Diaza-2,4-cyclopentadiene; Ondansetron EP Impurity E; Clotrimazole EP Impurity D
Enalapril EP Impurity I (Imidazole) is a fully characterized chemical compound used as a reference standard of API Enalapril. The standard offered is compliant with regulatory guidelines. Enalapril EP Impurity I (Imidazole) is used for analytical method development, method validation (AMV), and Quality Controlled (QC) applications during synthesis and formulation stages of drug development and serves as a reference standard for traceability against pharmacopeial standards (USP or EP). Axios Research products are intended for analytical purposes only and are not for human use. CAS - 288-32-4
Related products
Enalapril EP Impurity A Maleate ((S,S,R)-Enalapril Maleate)
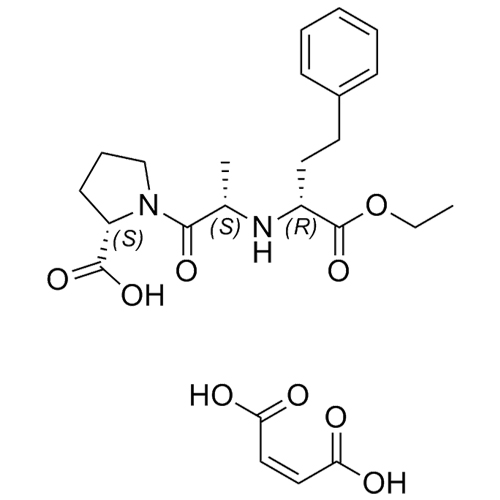
M.F.
M.W. 376.45 116.07
CAT# AR-E01327
CAS# 1356932-13-2
Enalapril EP Impurity H HCl (Mixture of Diastereomers)
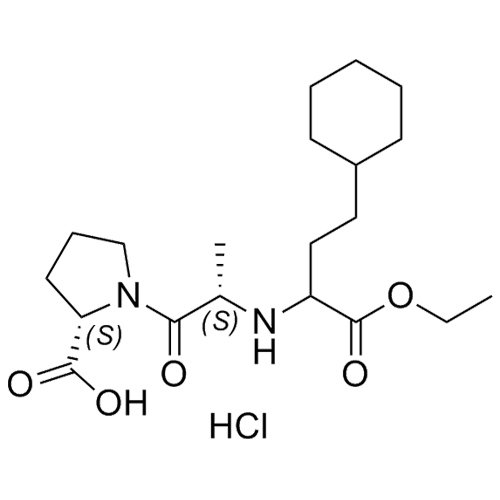
M.F.
M.W. 382.50 36.46
CAT# AR-E01332
CAS# NA
Enalapril EP Impurity D (Enalapril Diketopiperazine)
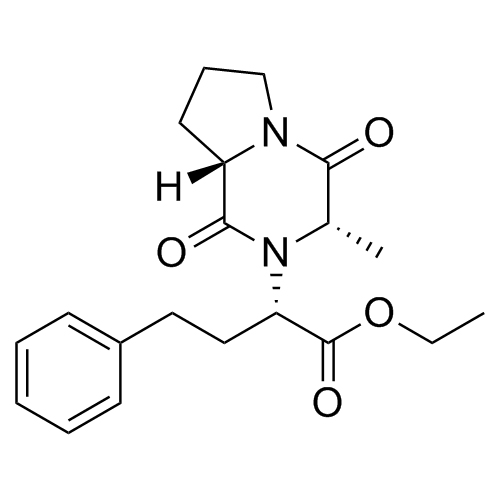
M.F.
M.W. 358.43
CAT# AR-E01330
CAS# 115729-52-7
Enalapril EP Impurity H HCl (Mixture of Diastereomers)
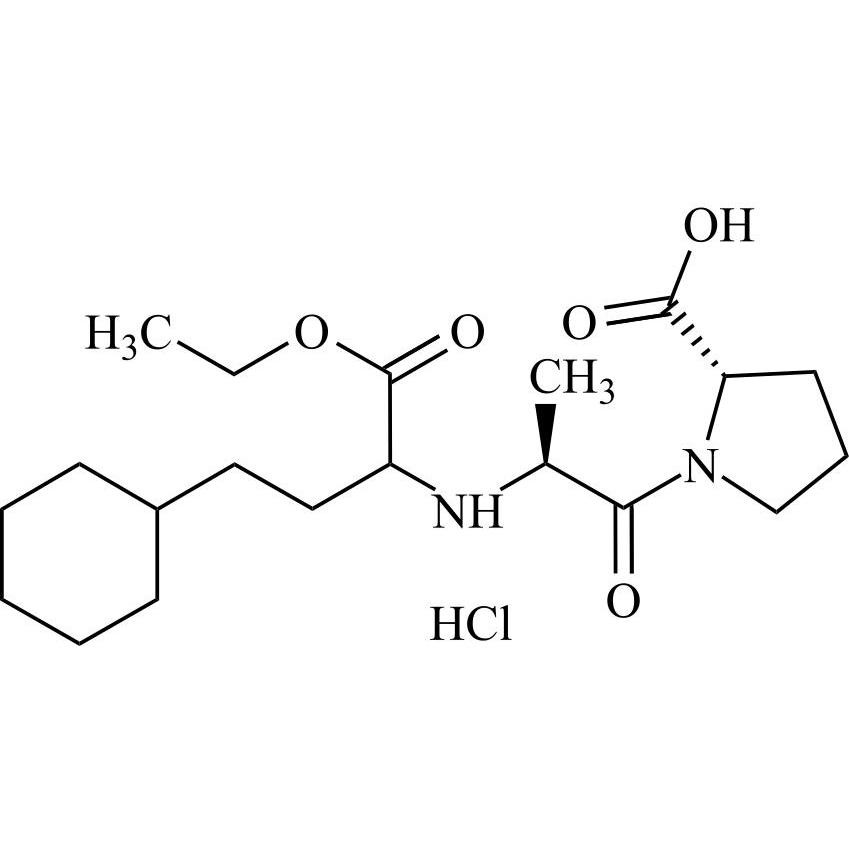
M.F.
M.W. 382.50 36.46
CAT# AR-E09799
CAS# NA
Enalapril Impurity 14 (Bis(2-chloroethyl)-Butanedioate)
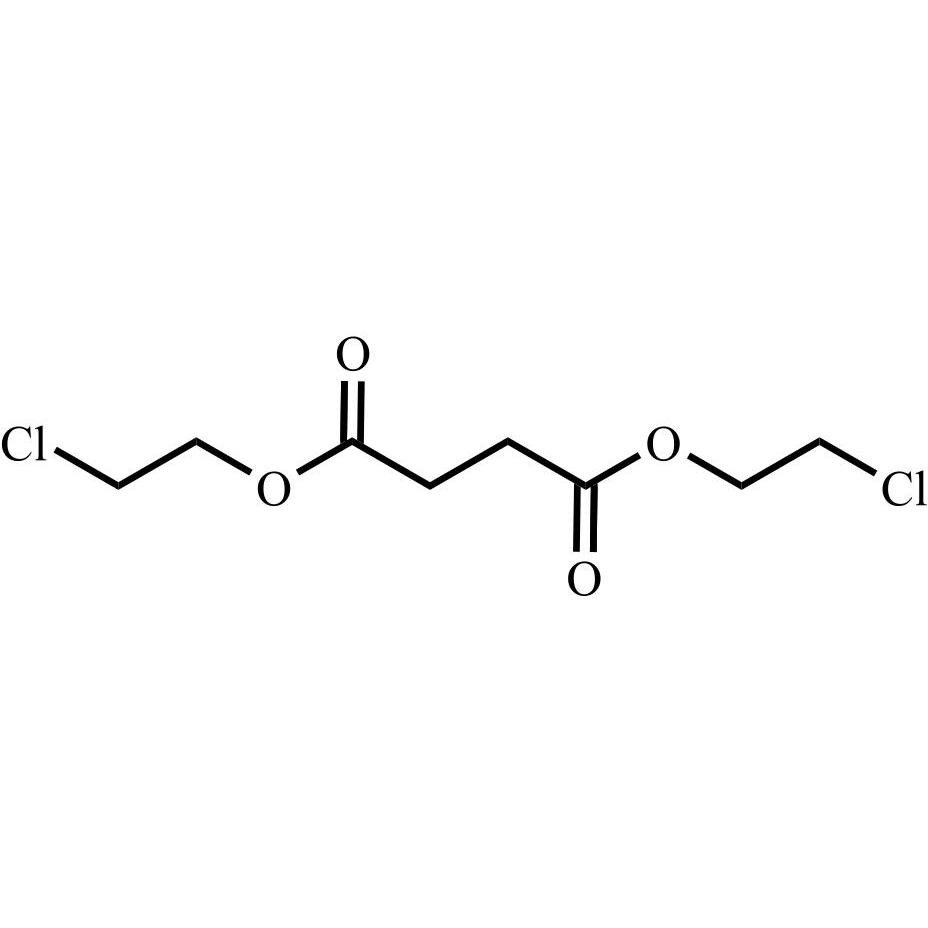
M.F.
M.W. 243.08
CAT# AR-E09795
CAS# 925-17-7













































