- Synonyms(1S,2R,3R,4S,5R,8R,9S,10S,11R,12R,14R)-11-(((2S,3R,4S,6R)-4-(dimethylamino)-3-hydroxy-6-methyltetrahydro-2H-pyran-2-yl)oxy)-5-ethyl-3-hydroxy-9-(((2R,4R,5S,6S)-5-hydroxy-4-methoxy-4,6-dimethyltetrahydro-2H-pyran-2-yl)oxy)-2,4,8,10,12,14-hexamethyl-6,15,16-trioxatricyclo[10.2.1.11,4]hexadecan-7-on...
- Description
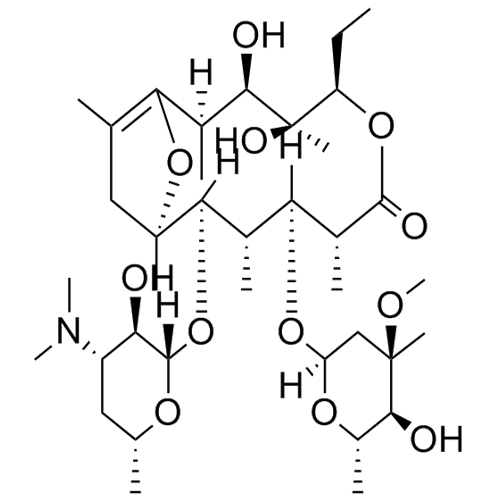
(1S,2R,3R,4S,5R,8R,9S,10S,11R,12R,14R)-11-(((2S,3R,4S,6R)-4-(dimethylamino)-3-hydroxy-6-methyltetrahydro-2H-pyran-2-yl)oxy)-5-ethyl-3-hydroxy-9-(((2R,4R,5S,6S)-5-hydroxy-4-methoxy-4,6-dimethyltetrahydro-2H-pyran-2-yl)oxy)-2,4,8,10,12,14-hexamethyl-6,15,16-trioxatricyclo[10.2.1.11,4]hexadecan-7-one; Anhydro Erythromycin A; 9-Deoxo-6,12-dideoxy-6,9:9,12-diepoxy-erythromycin; 6,9,12-Anhydroerythromycin A; Anhydroerythromycin; BRL 46355ER; EM 202; Erythromycin Anhydride, Impurity D; Erythromycin EP Impurity D
Erythromycin EP Impurity D (Anhydroerythromycin A) is a fully characterized chemical compound used as a reference standard of API Erythromycin. The standard offered is compliant with regulatory guidelines. Erythromycin EP Impurity D (Anhydroerythromycin A) is used for analytical method development, method validation (AMV), and Quality Controlled (QC) applications during synthesis and formulation stages of drug development and serves as a reference standard for traceability against pharmacopeial standards (USP or EP). Axios Research products are intended for analytical purposes only and are not for human use. CAS - 23893-13-2
Related products
Erythromycin EP Impurity E (Erythromycin A Enol Ether)
M.F.
M.W. 715.92
CAT# AR-E01814
CAS# 33396-29-1
2’,4’’-O-Bis(trimethylsilyl)-6-O-methylerythromycin A 9-[O-(1- methoxy-1-methylethyl) Oxime

M.F.
M.W. 979.45
CAT# AR-E01831
CAS# 119699-81-9
Erythromycin Impurity J (Roxithromycin EP Impurity C)
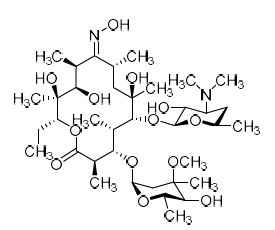
M.F.
M.W. 748.95
CAT# AR-E01801
CAS# 111321-02-9
Erythromycylamine-13C-d3 (Dirithromycin EP Impurity A-13C-d3)

M.F.
M.W. 738.98
CAT# AR-E10376
CAS# 26116-56-3 (non-labelled)
































































