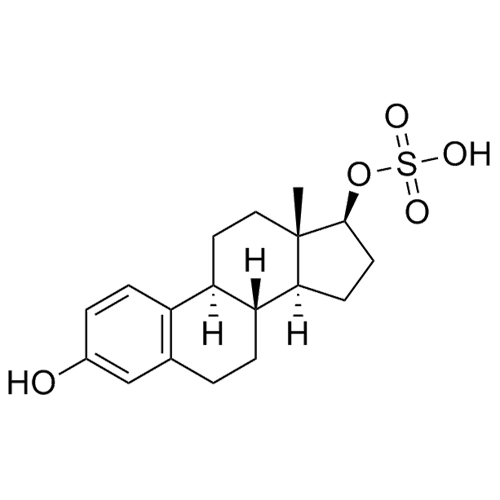
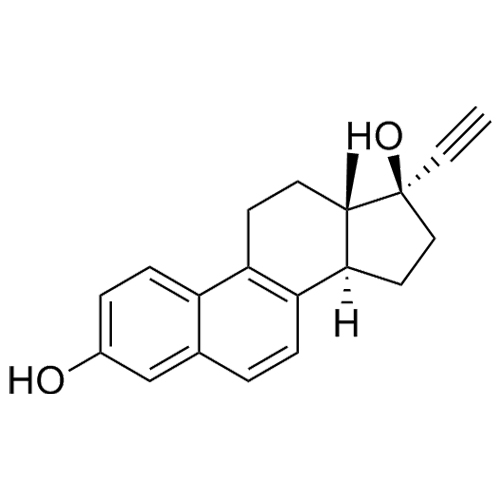
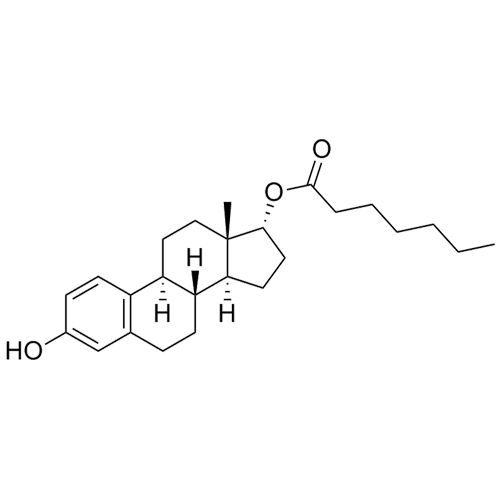
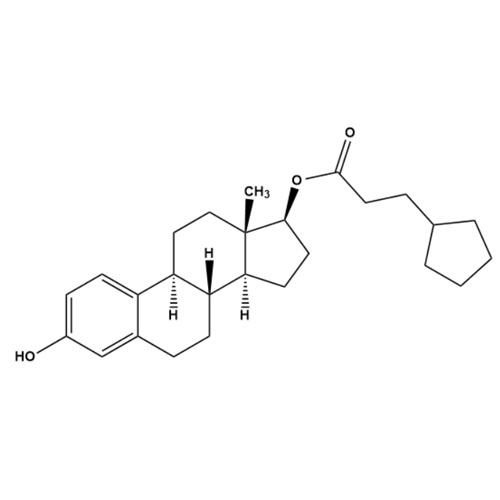
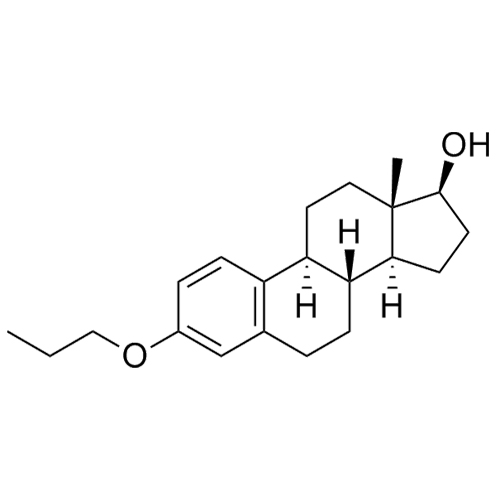
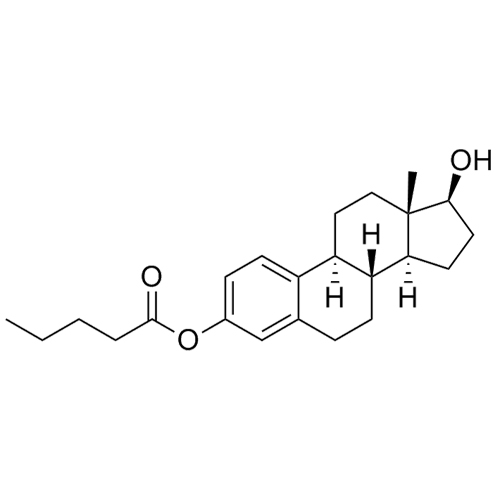
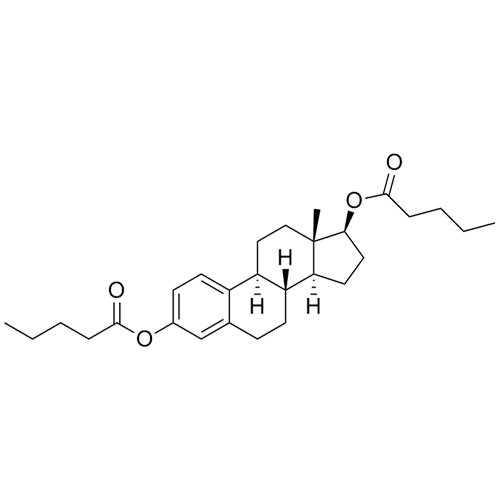
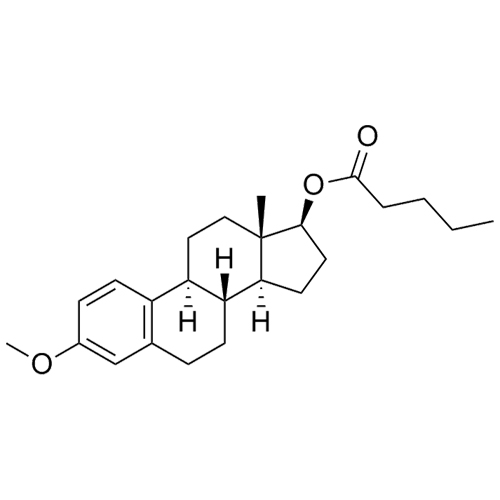
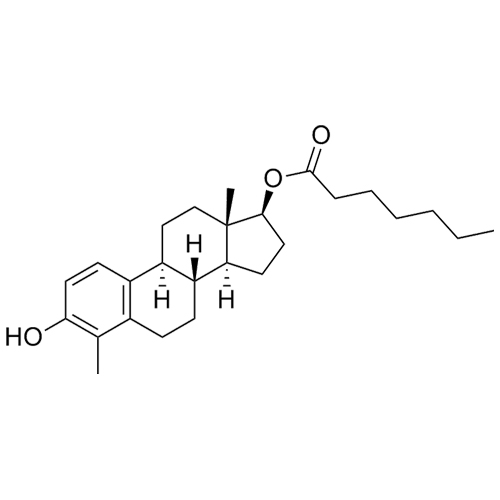
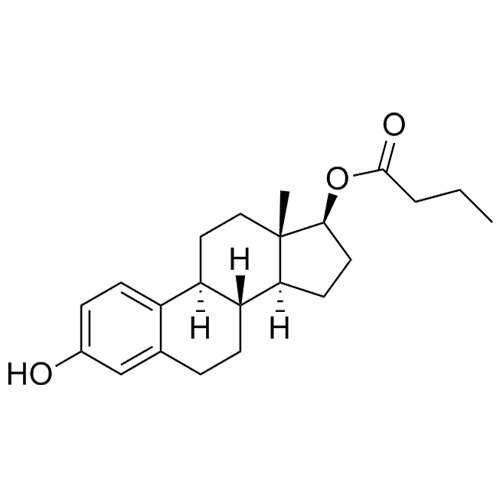
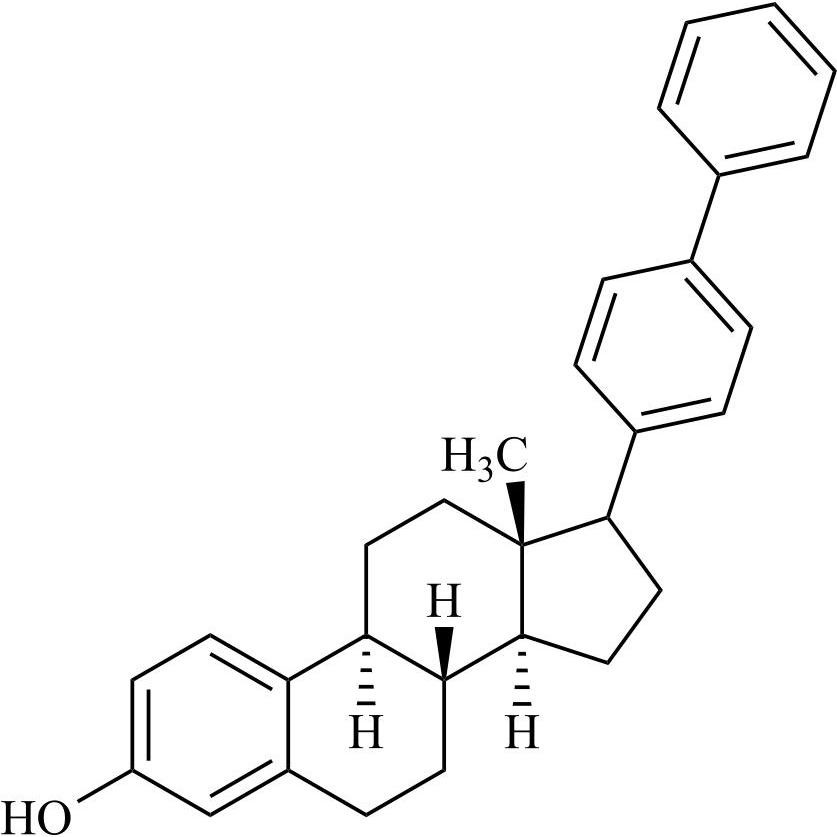
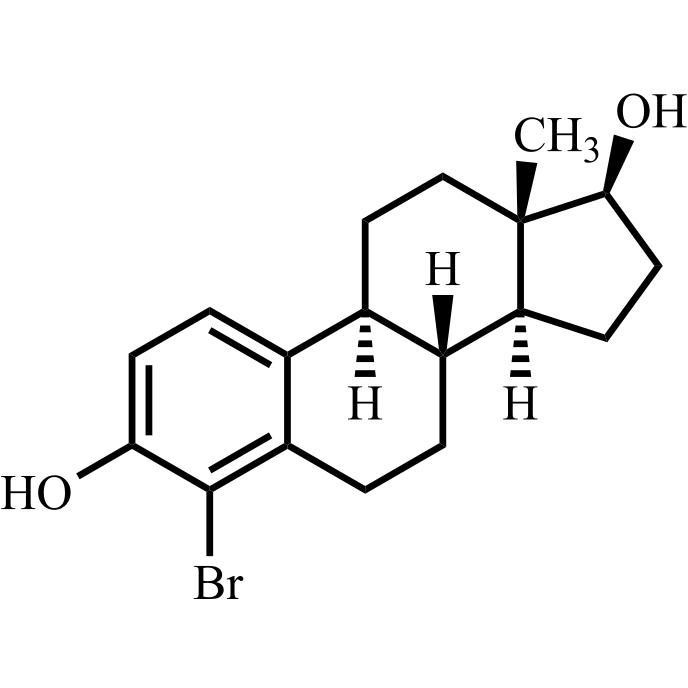
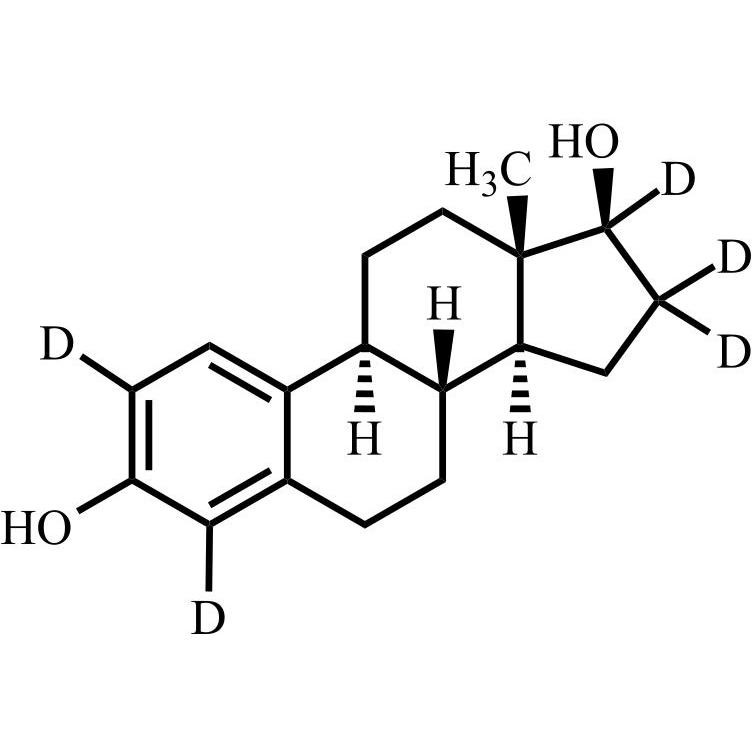
- Synonyms(8R,9S,13S,14S,17S)-3-hydroxy-13-methyl-2-pentanoyl-7,8,9,11,12,13,14,15,16,17-decahydro-6H-cyclopenta[a]phenanthren-17-yl pentanoate; 2-Valeryl-17?-estradiol 17-Valerate; (17?)-2-Valerylestra-1,3,5(10)-triene-1,17-diol 17-Pentanoate; 2-Valerylestradiol Valerate; 2-Valeryl-17-beta-Estradiol-17-Va...
- Description
(8R,9S,13S,14S,17S)-3-hydroxy-13-methyl-2-pentanoyl-7,8,9,11,12,13,14,15,16,17-decahydro-6H-cyclopenta[a]phenanthren-17-yl pentanoate; 2-Valeryl-17?-estradiol 17-Valerate; (17?)-2-Valerylestra-1,3,5(10)-triene-1,17-diol 17-Pentanoate; 2-Valerylestradiol Valerate; 2-Valeryl-17-beta-Estradiol-17-Valerate
Estradiol Valerate EP Impurity H is a fully characterized chemical compound used as a reference standard of API Estradiol. The standard offered is compliant with regulatory guidelines. Estradiol Valerate EP Impurity H is used for analytical method development, method validation (AMV), and Quality Controlled (QC) applications during synthesis and formulation stages of drug development and serves as a reference standard for traceability against pharmacopeial standards (USP or EP). Axios Research products are intended for analytical purposes only and are not for human use. CAS - 1421283-56-8
Related products
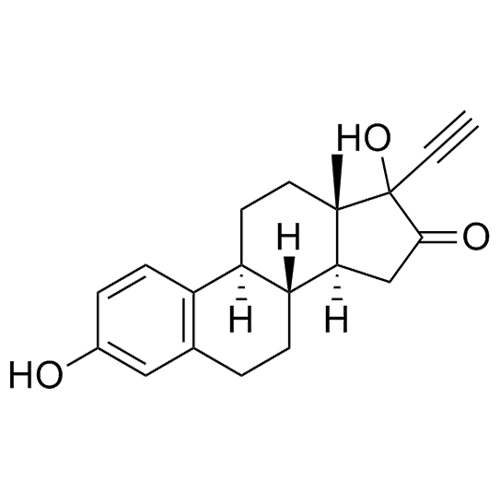
rac-Ethinylestradiol EP Impurity H (Mixture of Diastereomers)
M.F.
M.W. 310.39
CAT# AR-E01885
CAS# NA
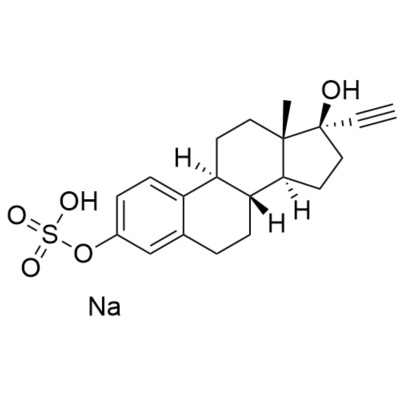
17-alpha-Ethynyl Estradiol-3-Sulfate (sodium salt)
M.F.
M.W. 376.48; 22.99
CAT# AR-E11085
CAS# 724762-79-2
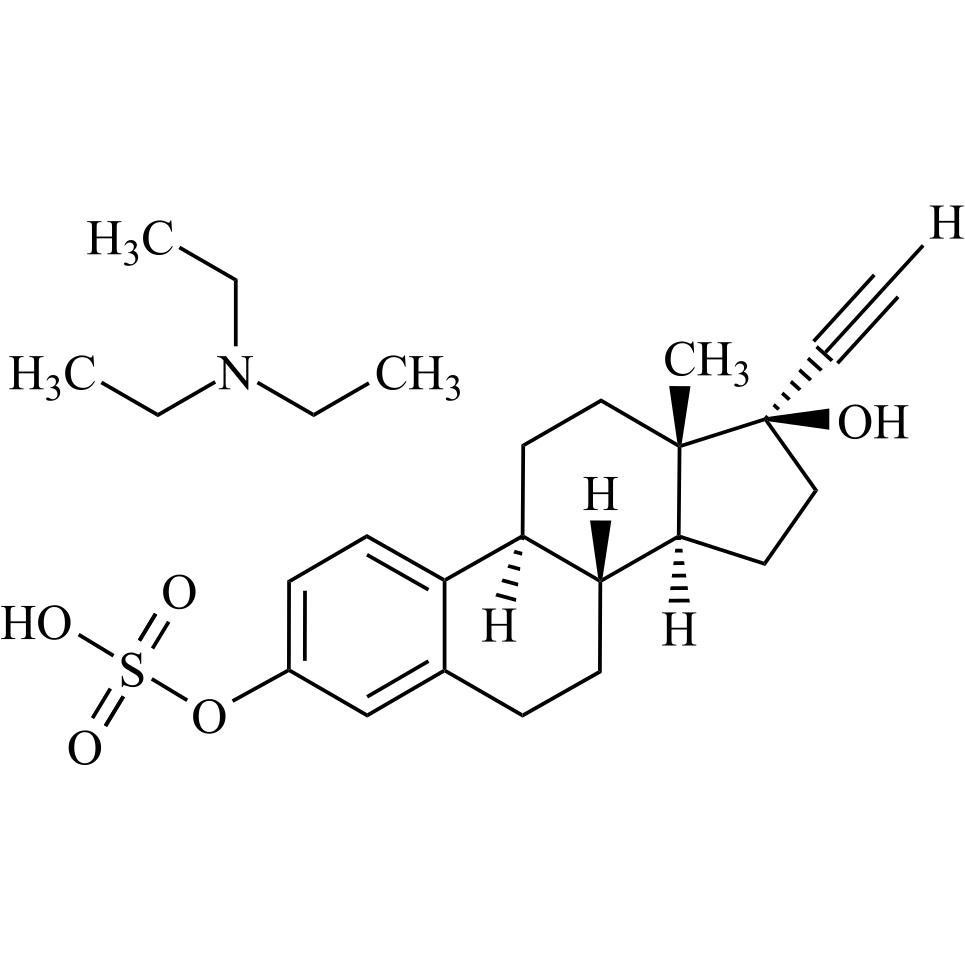
17-alpha-Ethynyl Estradiol-3-Sulfate Triethylamine Salt
M.F.
M.W. 376.48 101.19
CAT# AR-E10459
CAS# NA
17-alpha-Ethynyl Estradiol-17-Sulfate Triethylamine Salt
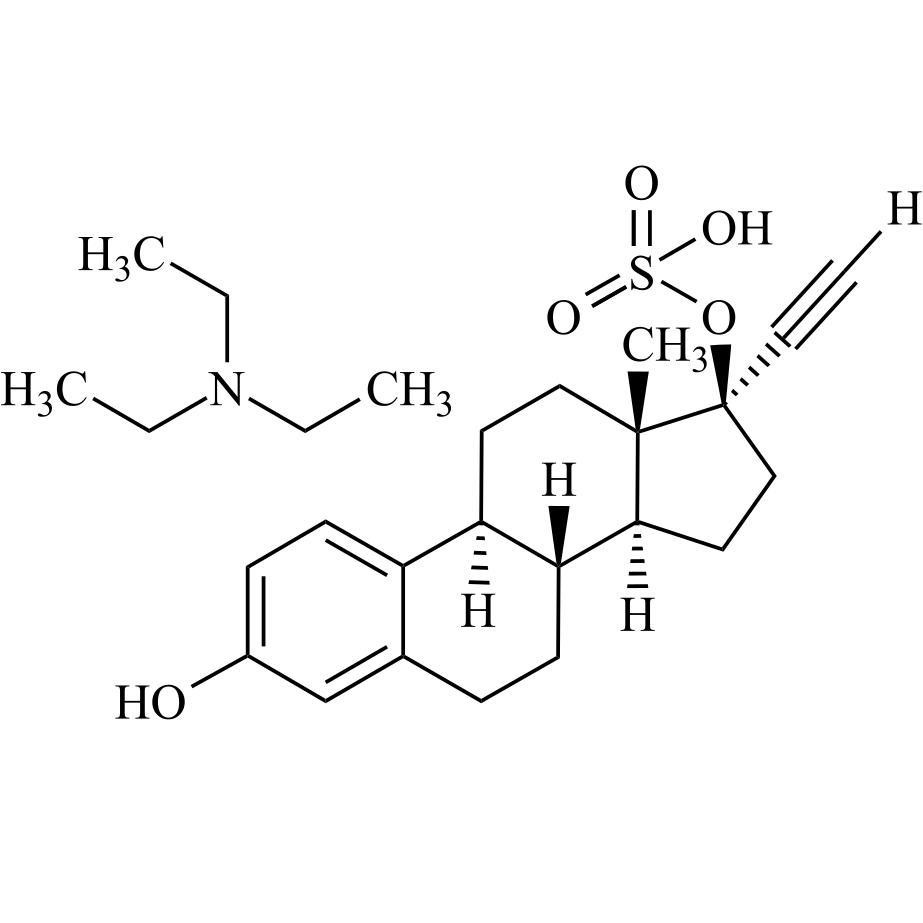
M.F.
M.W. 376.47 101.19
CAT# AR-E10464
CAS# NA
17-alpha-Ethynyl Estradiol-3-Sulfate-d7 Triethylamine Salt
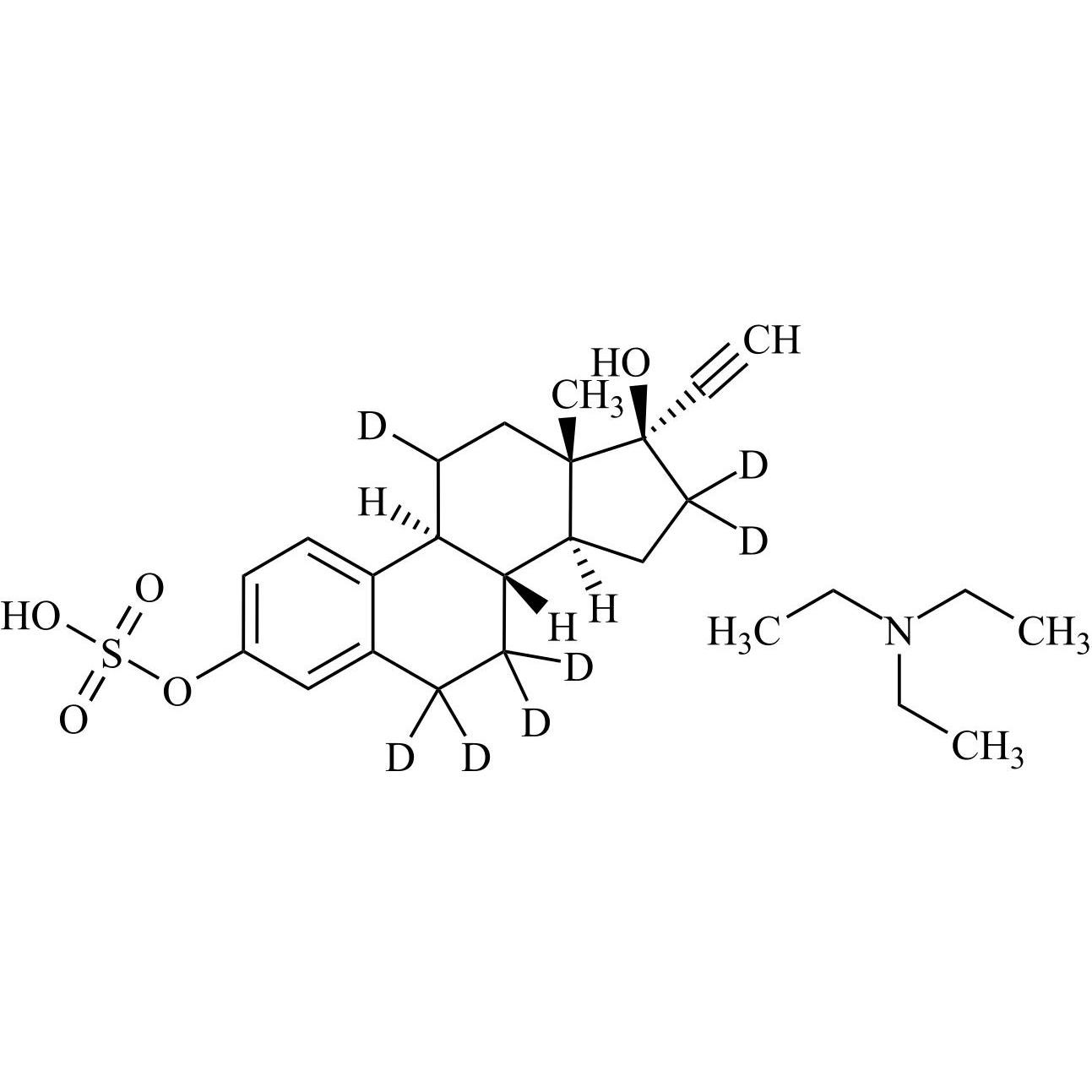
M.F.
M.W. 383.51 101.19
CAT# AR-E10452
CAS# NA
Estradiol Hemihydrate EP Impurity D-13C2 (delta-9(11)-Estradiol-13C2)
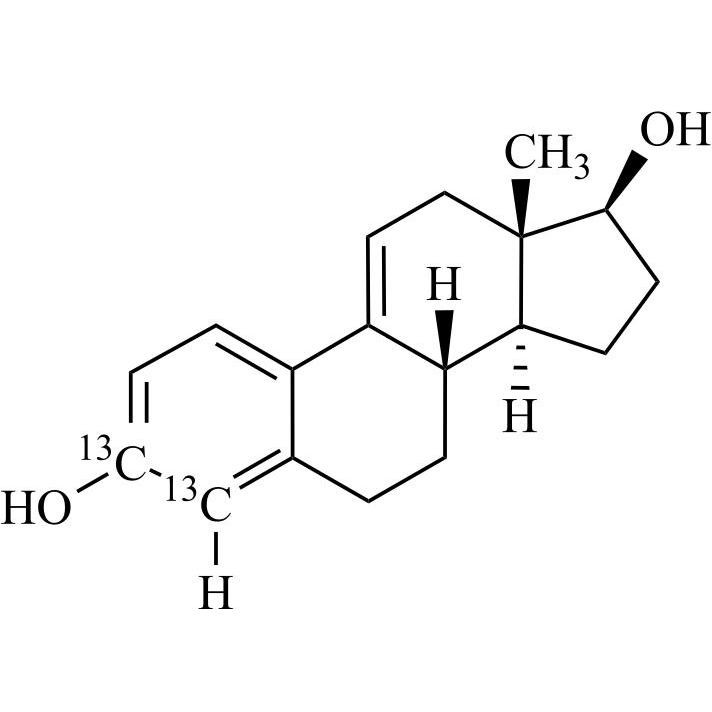
M.F.
M.W. 272.36
CAT# AR-E10435
CAS# NA
Estradiol-13C2-d2 (Estradiol Valerate EP Impurity A-13C2-d2, Estriol EP Impurity D-13C2-d2, Ethinylestradiol EP Impurity D-13C2-d2)
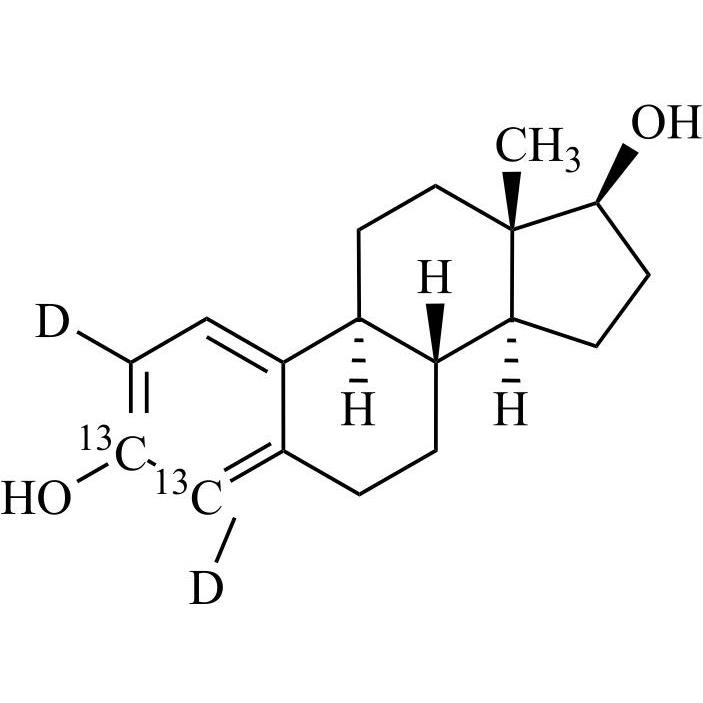
M.F.
M.W. 276.38
CAT# AR-E10458
CAS# NA
Estradiol Hemihydrate EP Impurity D-d3 (delta-9(11)-Estradiol-d3)
M.F.
M.W. 273.39
CAT# AR-E10462
CAS# NA