- Synonyms1-(4-isobutylphenyl)ethanone;1-[4-(2-Methylpropyl)phenyl]ethanone; 1-Acetyl-4-isobutylbenzene; 1-[4-(2-Methylpropyl)phenyl]-1-ethanone; 1-[4-(2-Methylpropyl)phenyl]ethanone; 4-(2-Methylpropyl)acetophenone; NSC 173015; p-Acetylisobutylbenzene; p-Isobutylacetophenone; p-Isobutylphenyl Methyl Ketone...
- Description
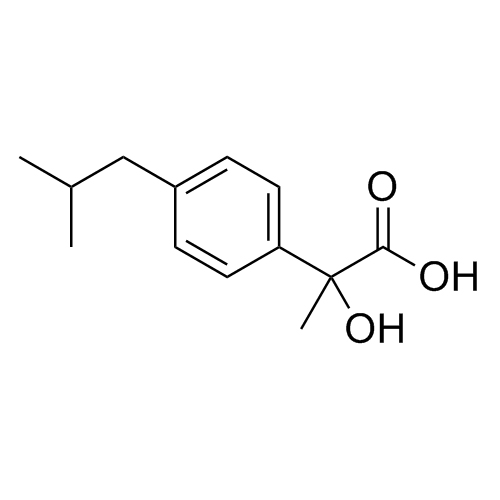
1-(4-isobutylphenyl)ethanone;1-[4-(2-Methylpropyl)phenyl]ethanone; 1-Acetyl-4-isobutylbenzene; 1-[4-(2-Methylpropyl)phenyl]-1-ethanone; 1-[4-(2-Methylpropyl)phenyl]ethanone; 4-(2-Methylpropyl)acetophenone; NSC 173015; p-Acetylisobutylbenzene; p-Isobutylacetophenone; p-Isobutylphenyl Methyl Ketone; 4-IBAP; USP Ibuprofen Related Compound C; Ibuprofen EP Impurity E;4'-Isobutylacetophenone
Ibuprofen EP Impurity E is a fully characterized chemical compound used as a reference standard of API Ibuprofen. The standard offered is compliant with regulatory guidelines. Ibuprofen EP Impurity E is used for analytical method development, method validation (AMV), and Quality Controlled (QC) applications during synthesis and formulation stages of drug development and serves as a reference standard for traceability against pharmacopeial standards (USP or EP). Axios Research products are intended for analytical purposes only and are not for human use. CAS - 38861-78-8
Related products
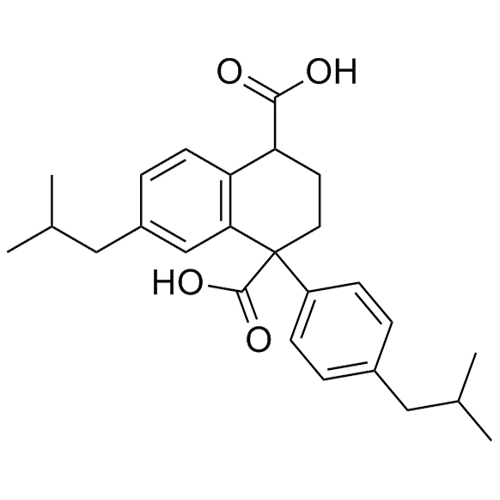
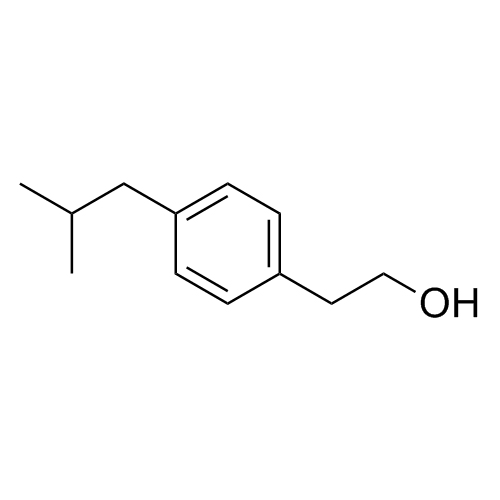
1,3-Dihydroxyprop-2-yl 2-(4-Isobutylphenyl)Propanonate

M.F.
M.W. 280.37
CAT# AR-I01067
CAS# 124635-85-4
2-(1-Chloroethyl)-2-(4′-isobutylphenyl)-5,5-dimethyl-1,3-dioxane

M.F.
M.W. 310.86
CAT# AR-I01113
CAS# 89878-72-8
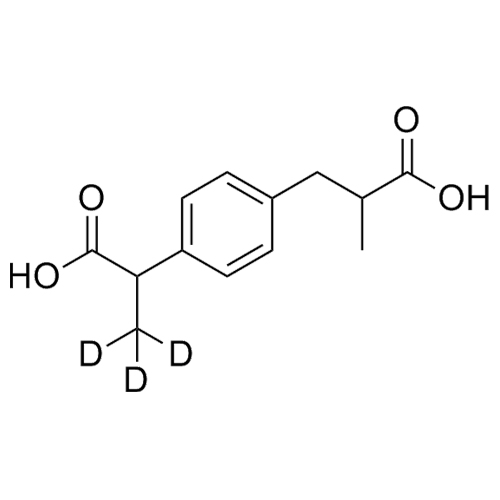
Ibuprofen Carboxylic Acid-d3 (Mixture of Diastereomers)
M.F.
M.W. 239.29
CAT# AR-I01110
CAS# 1216505-29-1
Ibuprofen Acyl Glucuronide (Mixture of Diastereomers)

M.F.
M.W. 382.41
CAT# AR-I01043
CAS# 115075-59-7