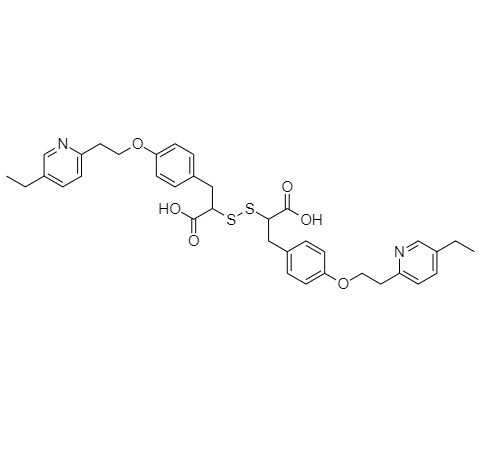
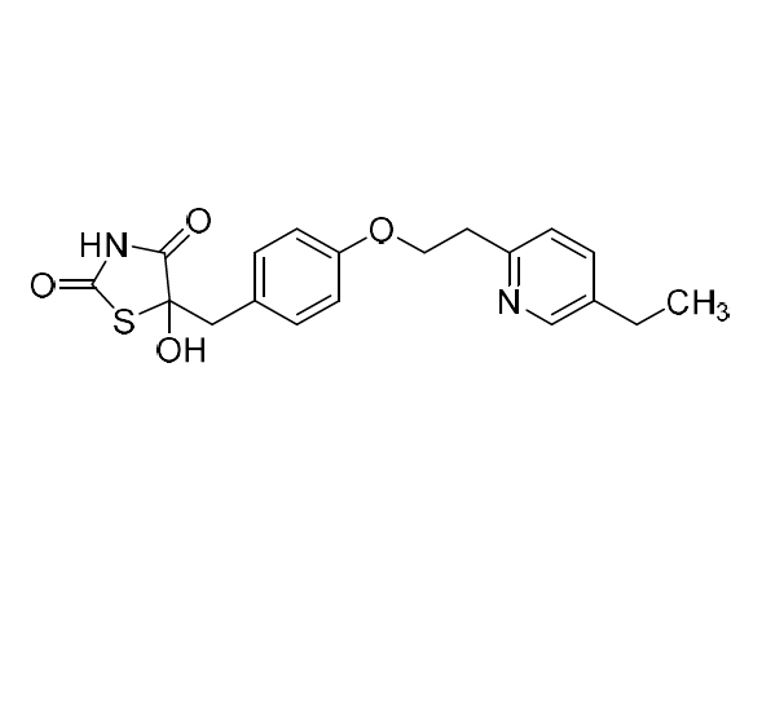
5-(4-fluorobenzyl)thiazolidine-2,4-dione
Pioglitazone Fluoro Impurity is a fully characterized chemical compound used as a reference standard of API Pioglitazone. The standard offered is compliant with regulatory guidelines. Pioglitazone Fluoro Impurity is used for analytical method development, method validation (AMV), and Quality Controlled (QC) applications during synthesis and formulation stages of drug development and serves as a reference standard for traceability against pharmacopeial standards (USP or EP). Axios Research products are intended for analytical purposes only and are not for human use. CAS - N/A
Related products
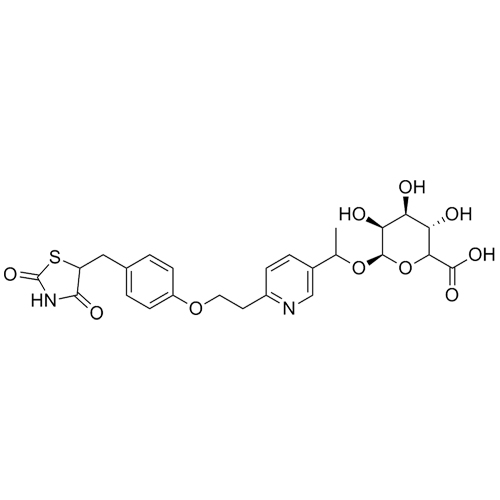
Pioglitazone N-beta-D-Glucuronide (Mixture of Diastereomers)

M.F.
M.W. 532.57
CAT# AR-P02014
CAS# 1296832-75-1