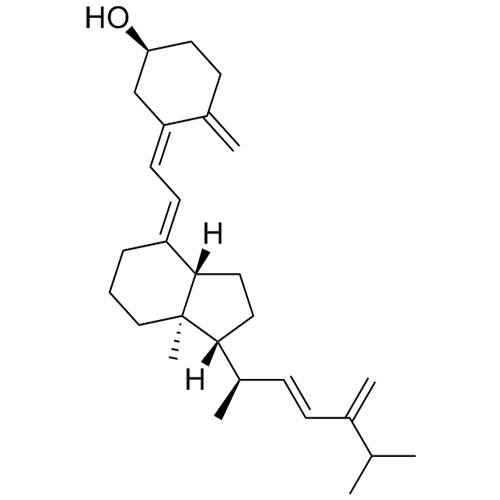
(3S,9S,10R,13R,14R,17R)-17-((2R,5R,E)-5,6-dimethylhept-3-en-2-yl)-10,13-dimethyl-2,3,4,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-3-ol; (22E)-Ergosta-5,7,22-trien-3β-ol; Ergosterol
Ergocalciferol EP Impurity B (Ergosterol) is a fully characterized chemical compound used as a reference standard of API Ergocalciferol. The standard offered is compliant with regulatory guidelines. Ergocalciferol EP Impurity B (Ergosterol) is used for analytical method development, method validation (AMV), and Quality Controlled (QC) applications during synthesis and formulation stages of drug development and serves as a reference standard for traceability against pharmacopeial standards (USP or EP). Axios Research products are intended for analytical purposes only and are not for human use. CAS - 57-87-4