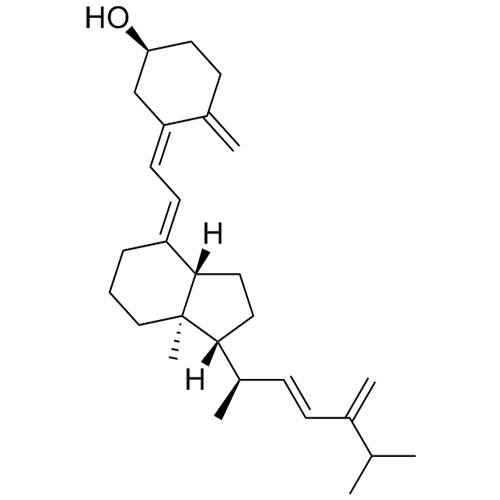
- Synonyms(S)-3-((E)-2-((1R,3aR,7aR)-1-((2R,5R,E)-5,6-dimethylhept-3-en-2-yl)-7a-methyl-2,3,3a,6,7,7a-hexahydro-1H-inden-4-yl)vinyl)-4-methylcyclohex-3-en-1-ol
- Description
(S)-3-((E)-2-((1R,3aR,7aR)-1-((2R,5R,E)-5,6-dimethylhept-3-en-2-yl)-7a-methyl-2,3,3a,6,7,7a-hexahydro-1H-inden-4-yl)vinyl)-4-methylcyclohex-3-en-1-ol
Ergocalciferol EP Impurity E (Tachysterol) is a fully characterized chemical compound used as a reference standard of API Ergocalciferol. The standard offered is compliant with regulatory guidelines. Ergocalciferol EP Impurity E (Tachysterol) is used for analytical method development, method validation (AMV), and Quality Controlled (QC) applications during synthesis and formulation stages of drug development and serves as a reference standard for traceability against pharmacopeial standards (USP or EP). Axios Research products are intended for analytical purposes only and are not for human use. CAS - 115-61-7
Related products
Ergocalciferol (Vitamin D2) EP Impurity A (5,6-trans-Vitamin D2)

M.F.
M.W. 396.66
CAT# AR-V01599
CAS# 51744-66-2
Ergocalciferol (Vitamin D2) Impurity 1 (Windaus Ketone)

M.F.
M.W. 276.47
CAT# AR-V01600
CAS# 55812-80-1