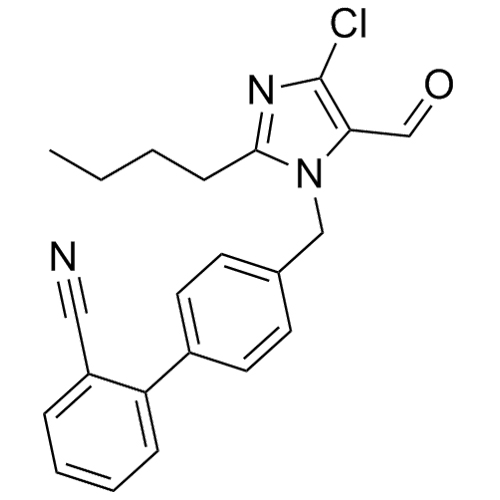
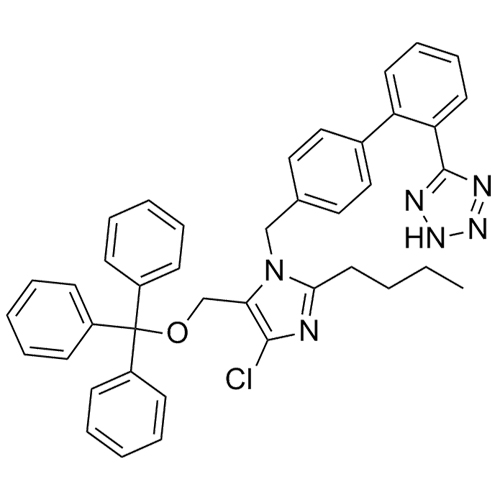
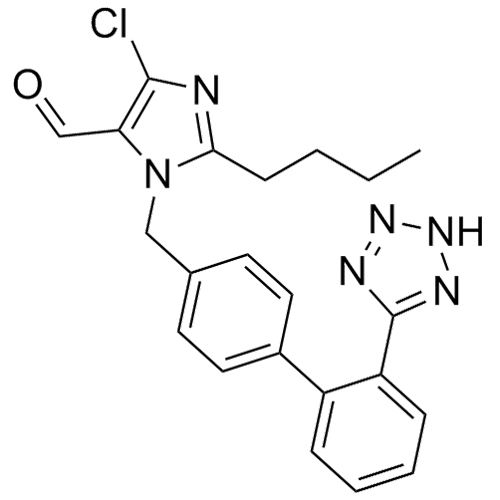
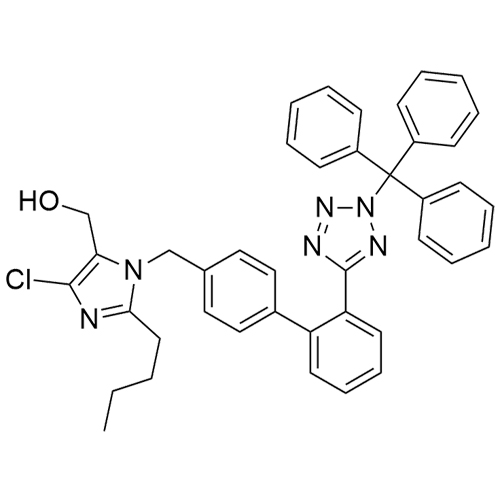
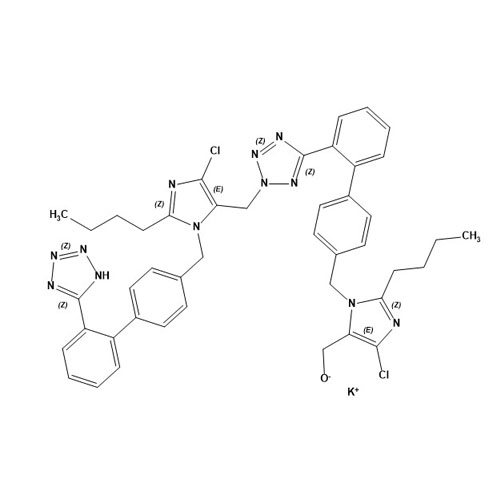
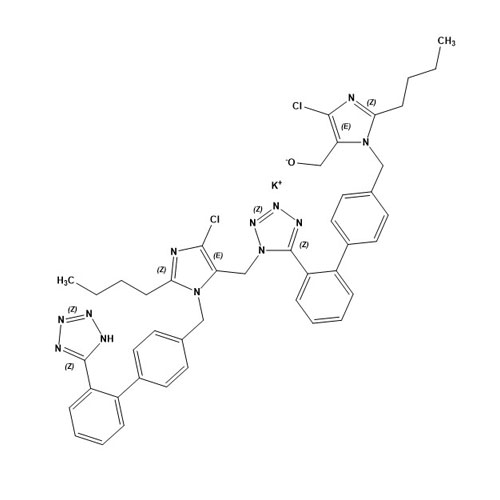
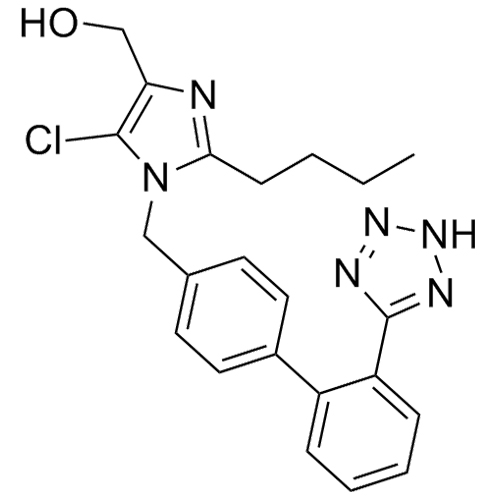
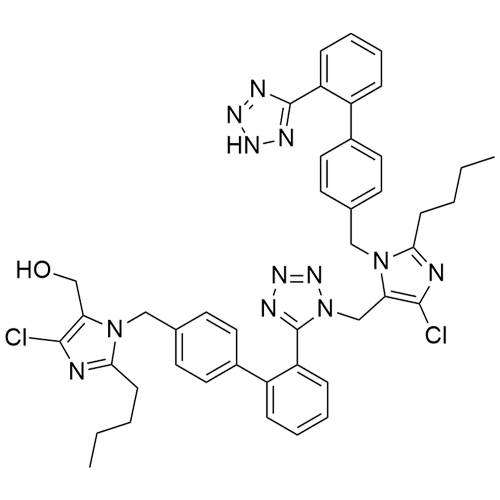
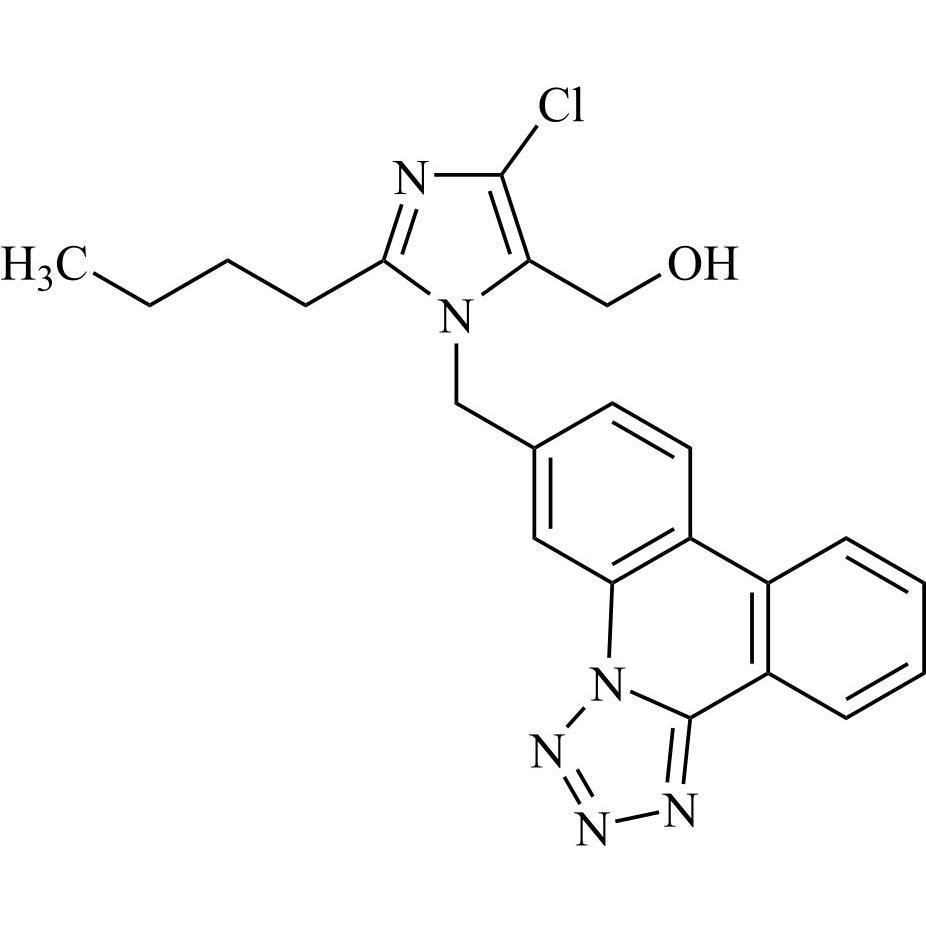
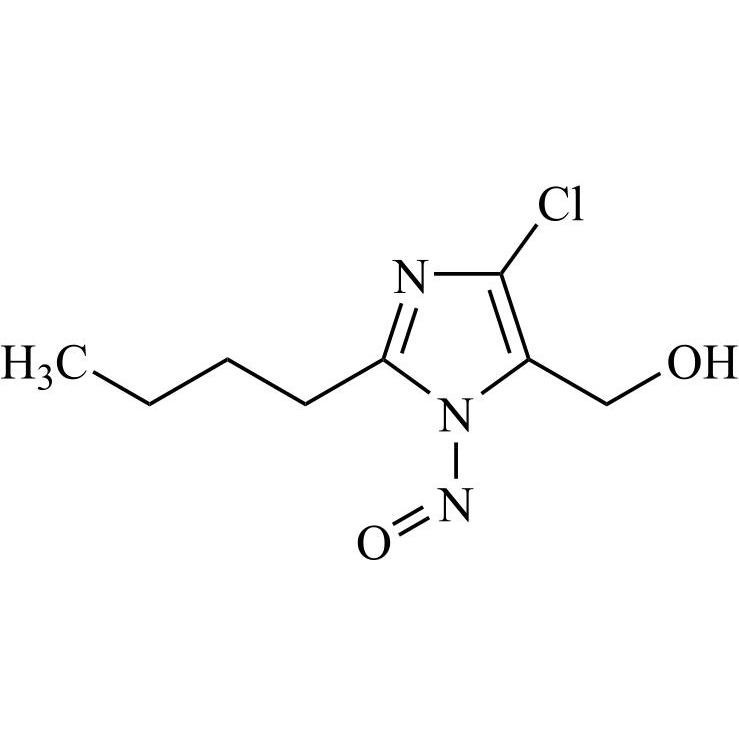
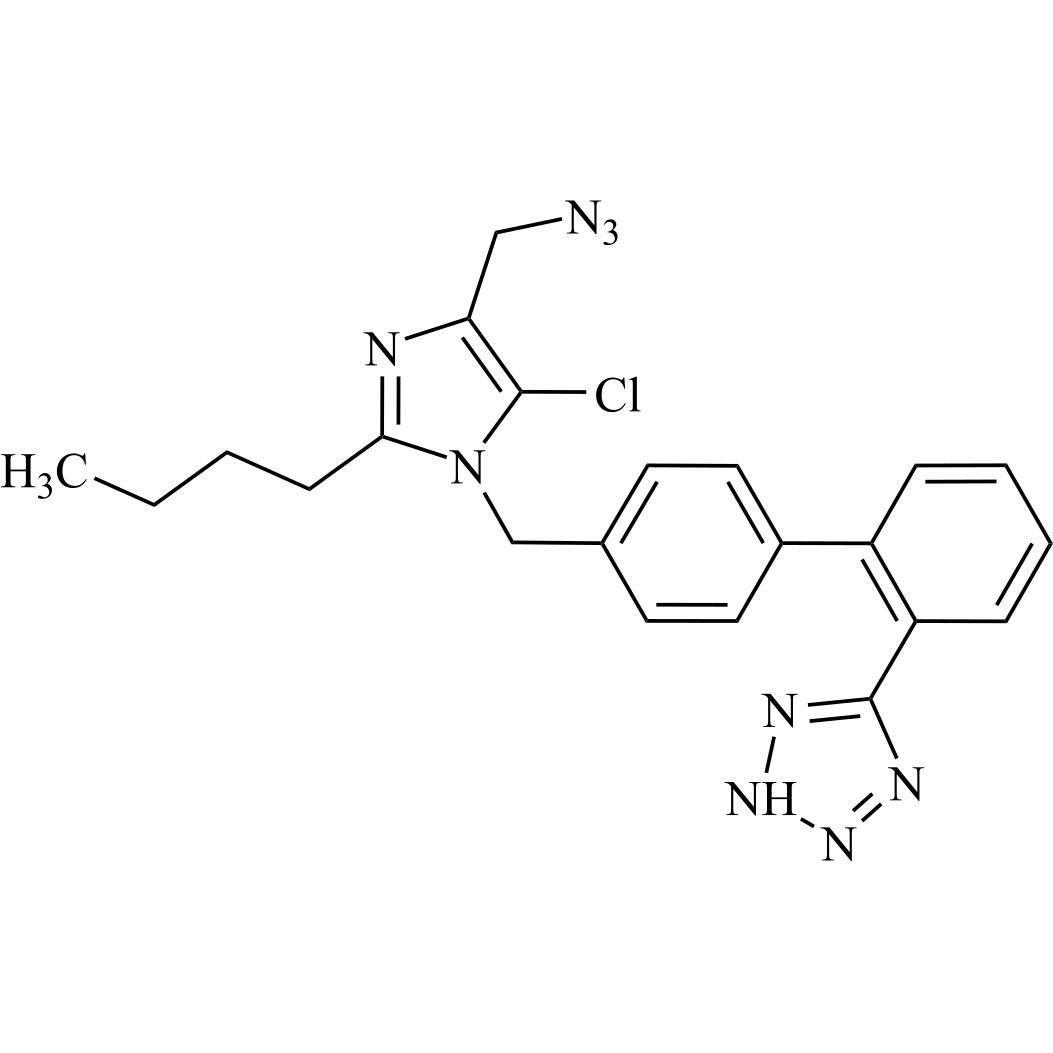
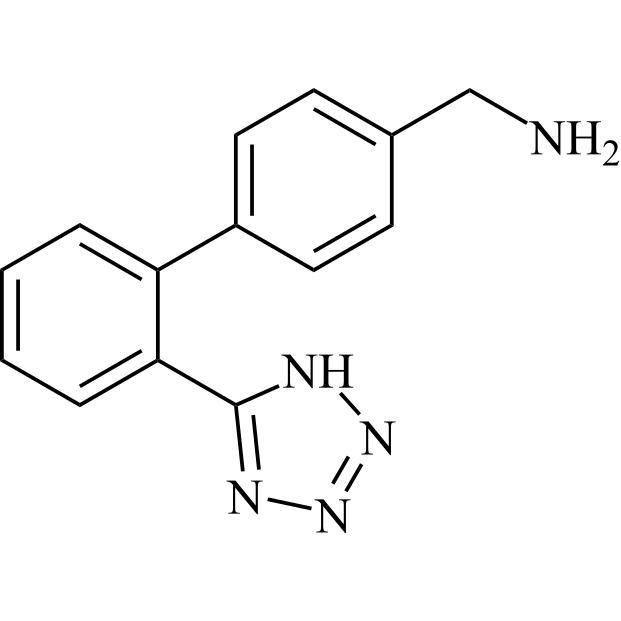
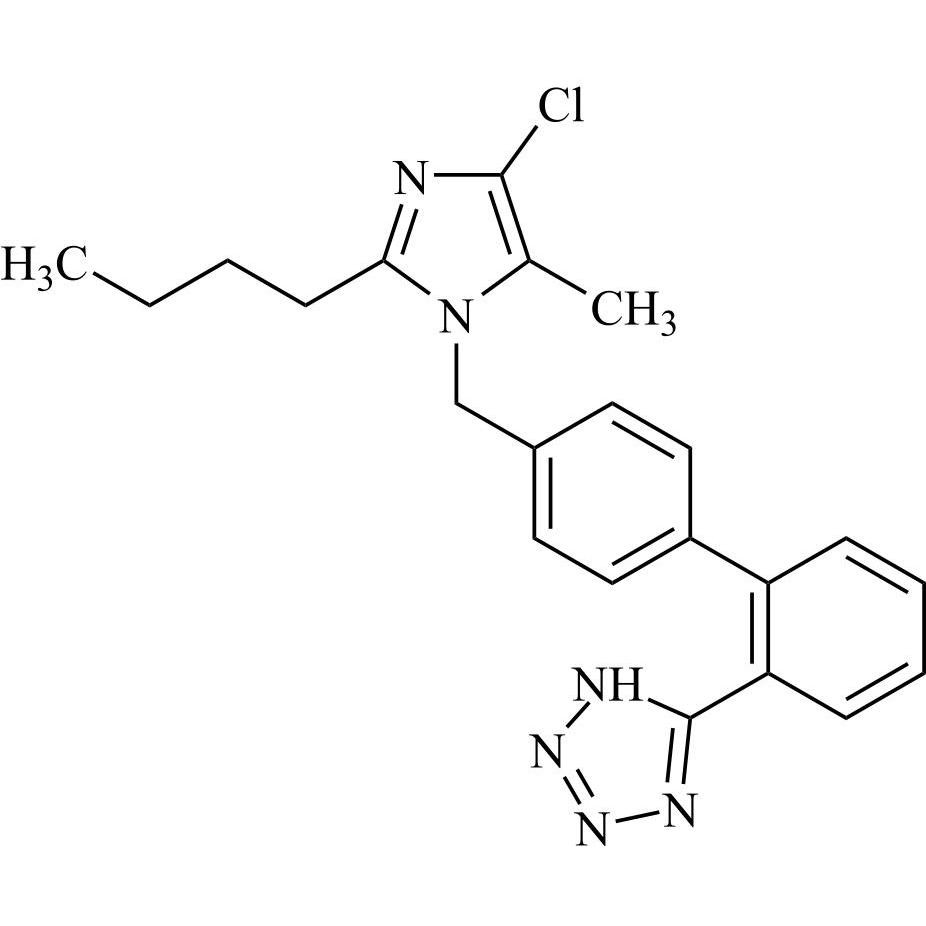
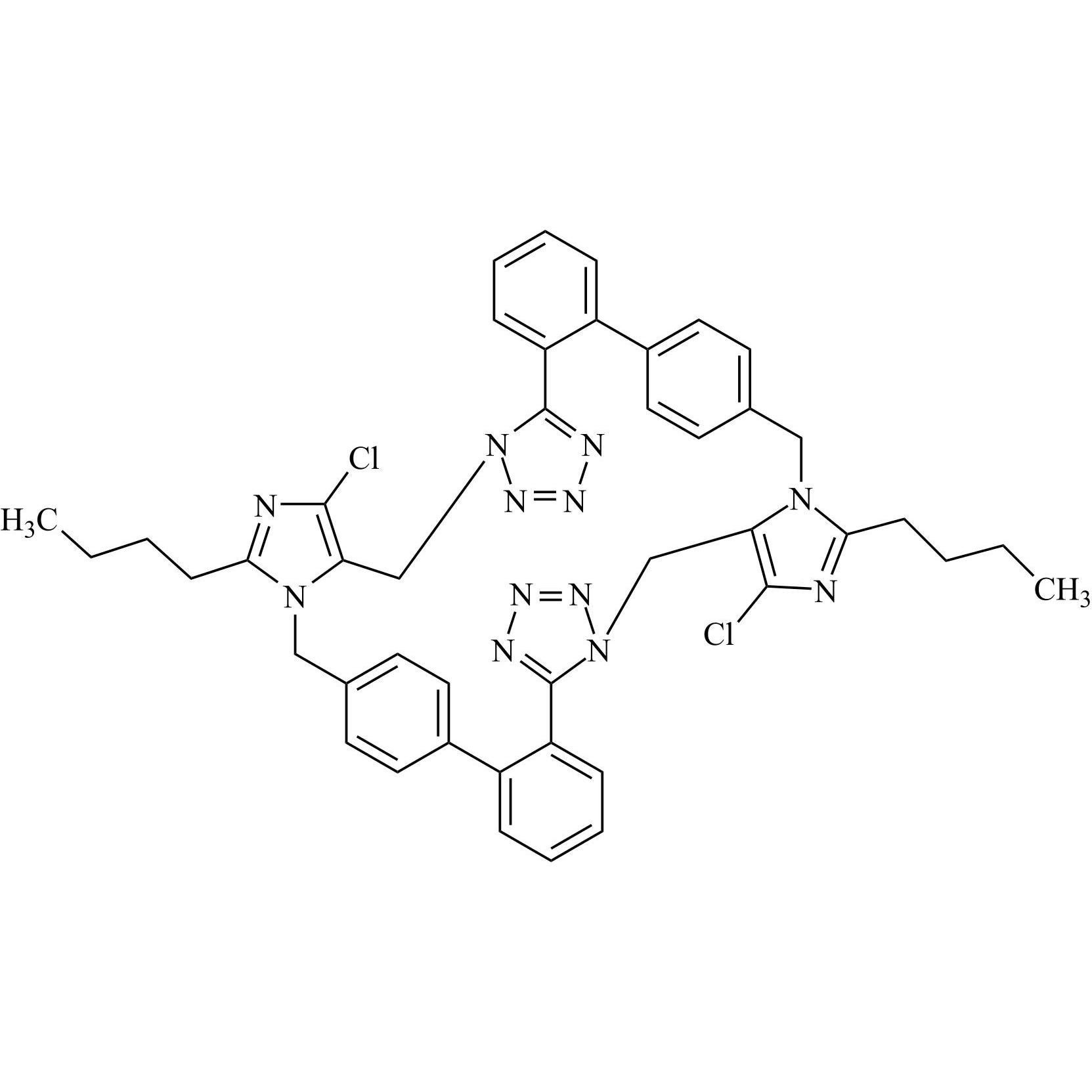
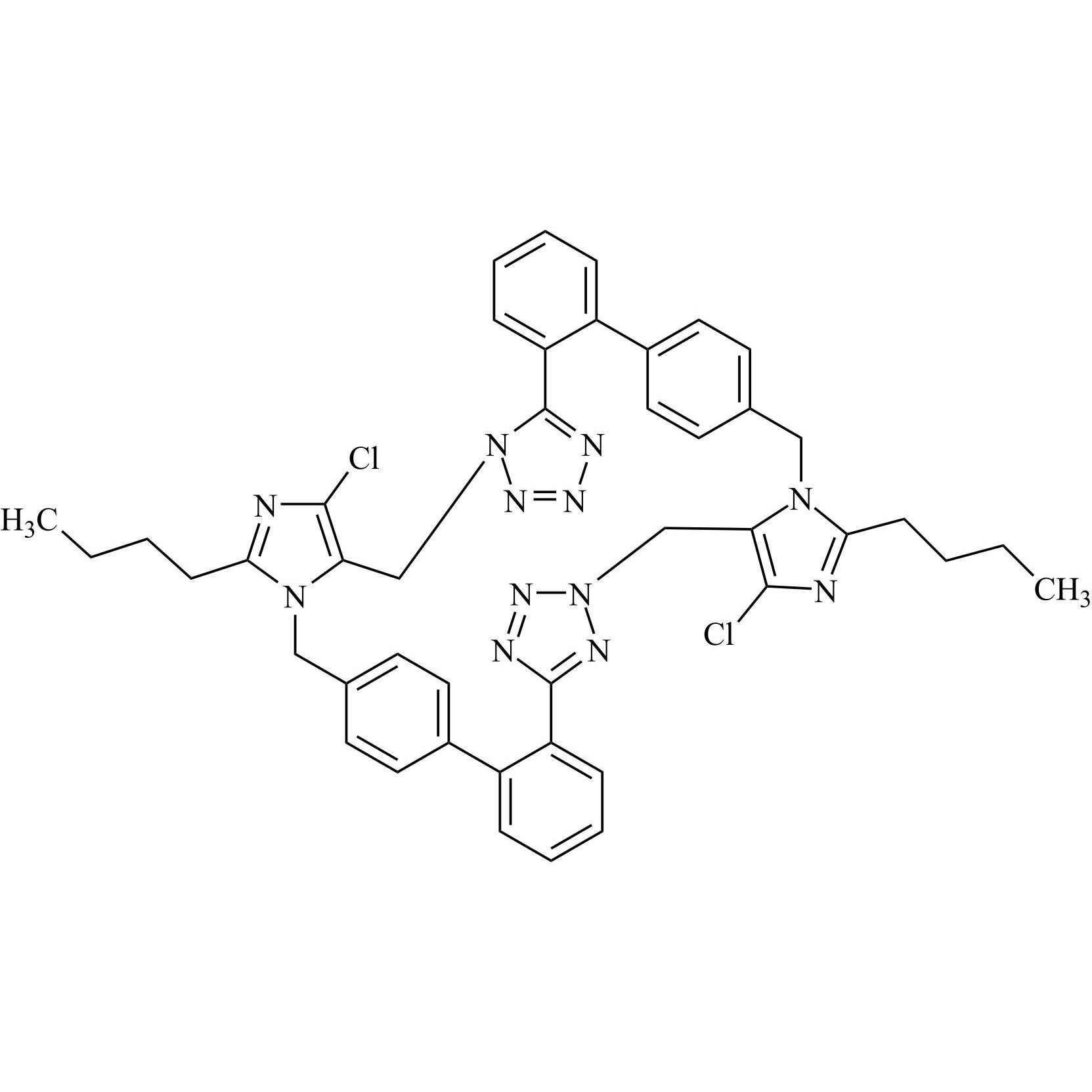
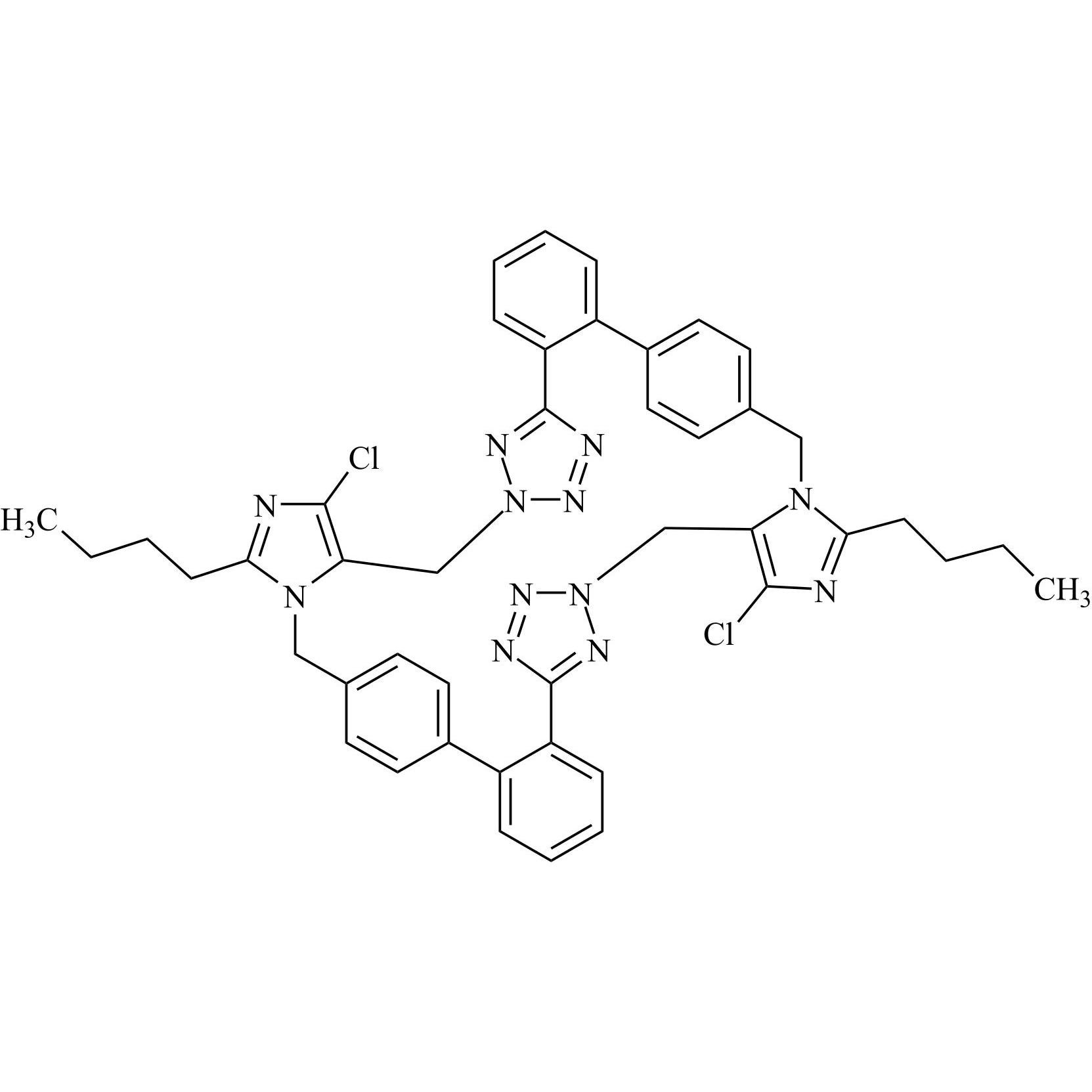
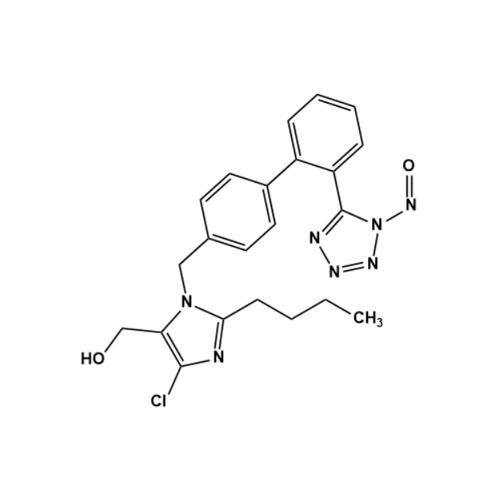
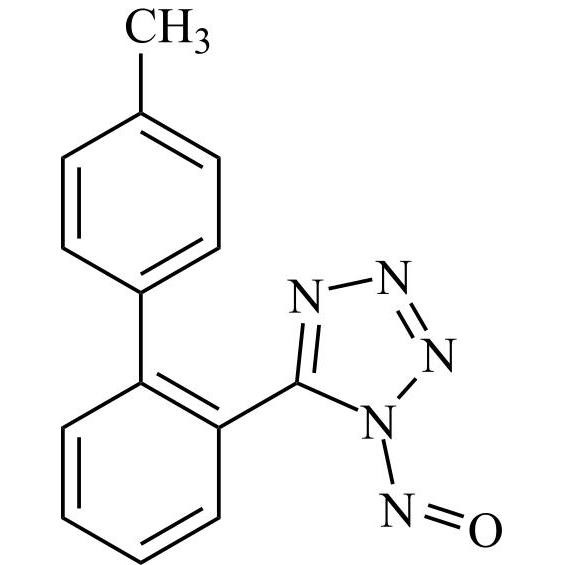
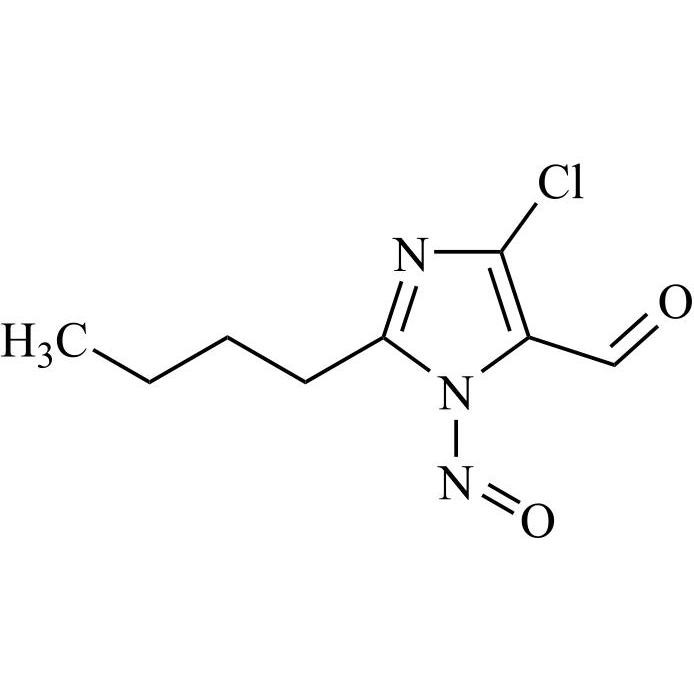
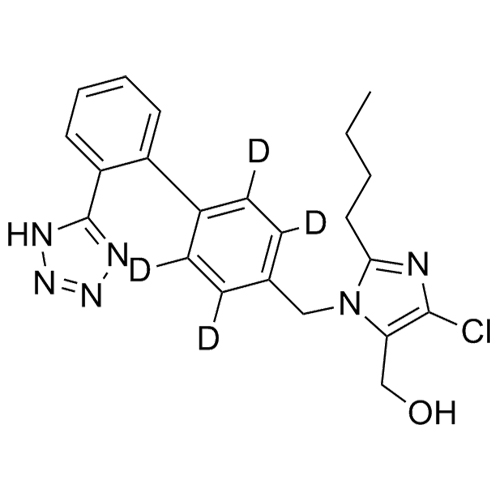
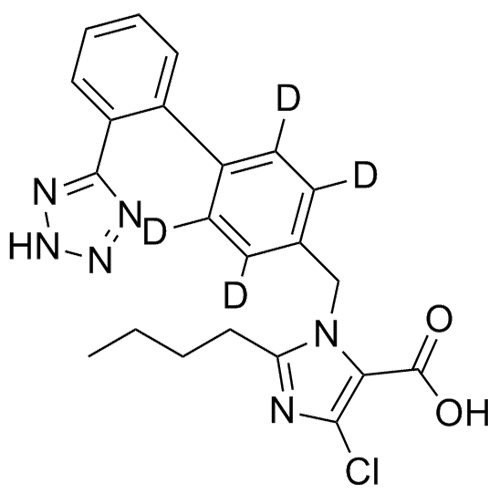
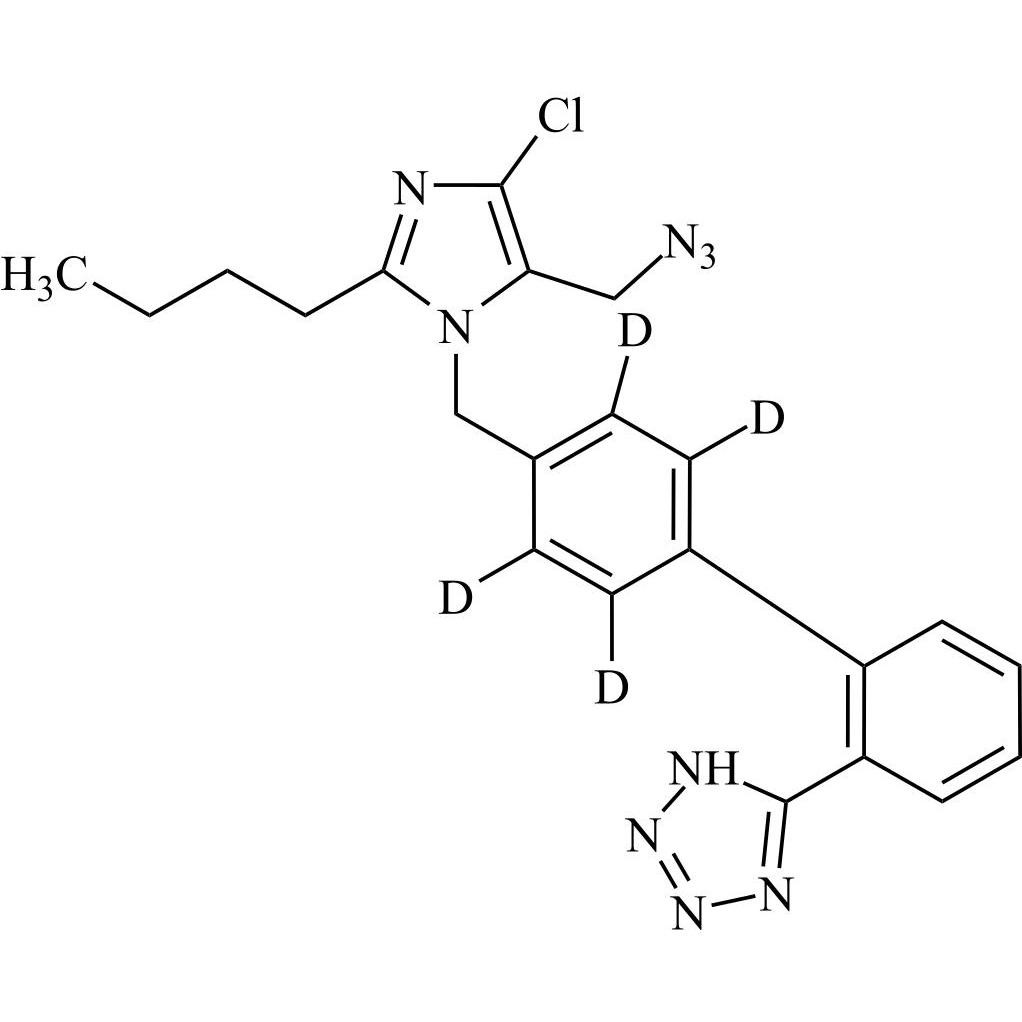
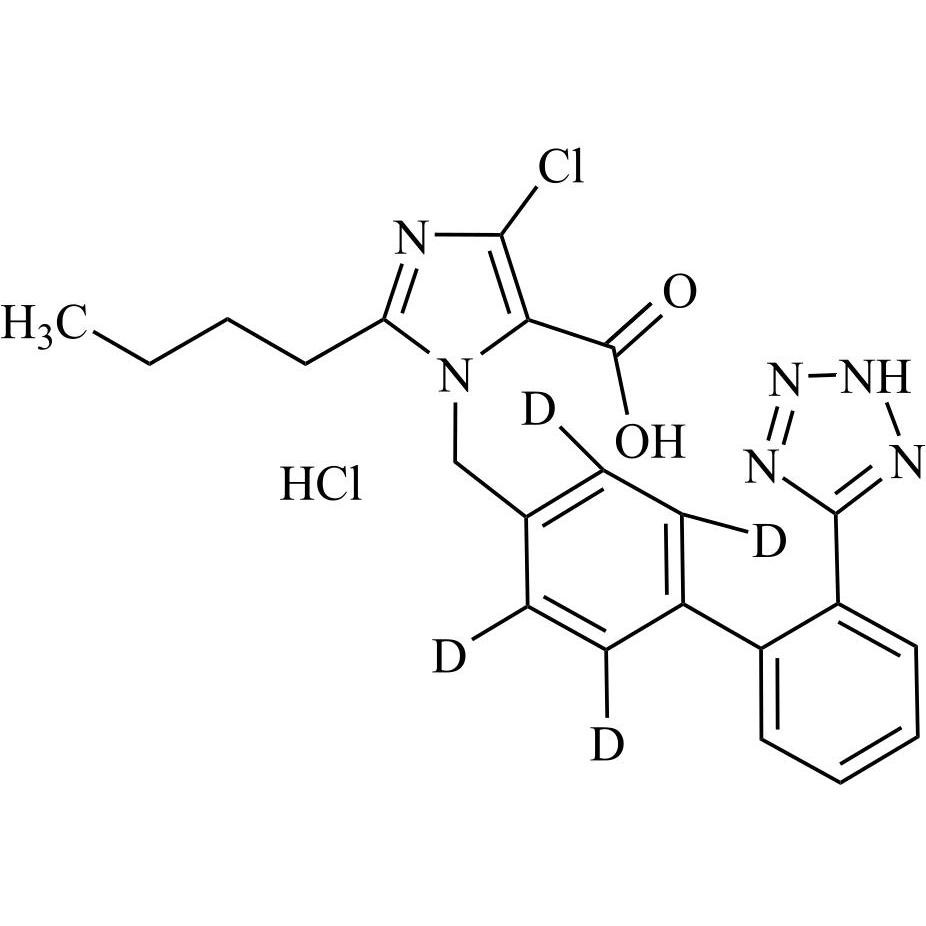
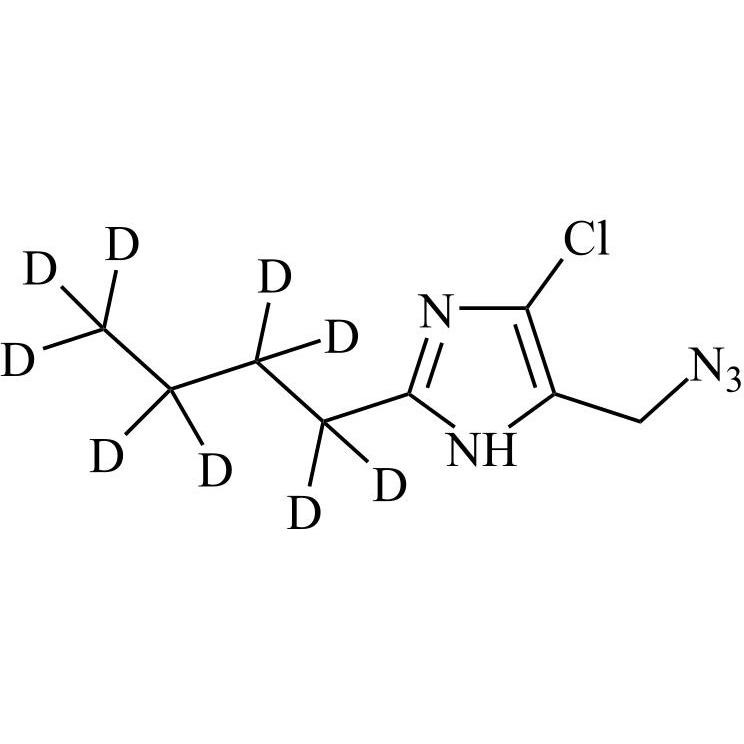
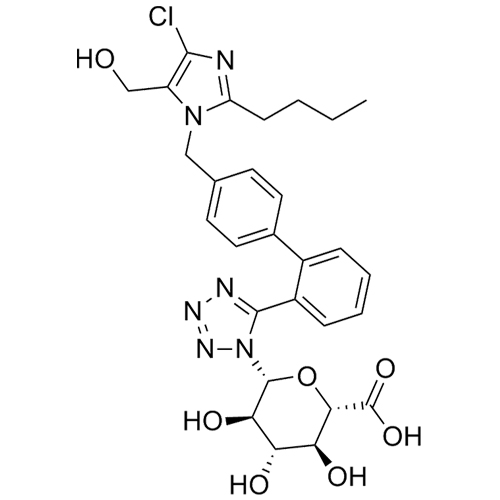
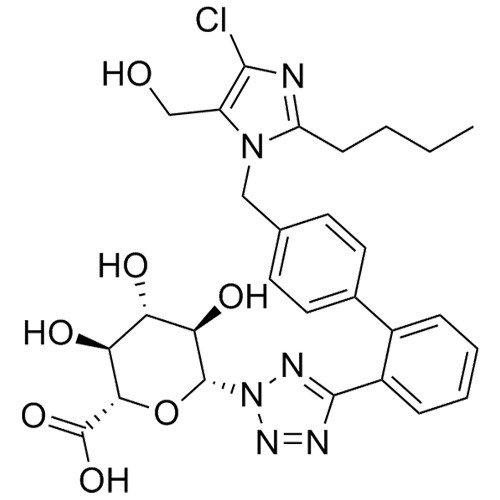
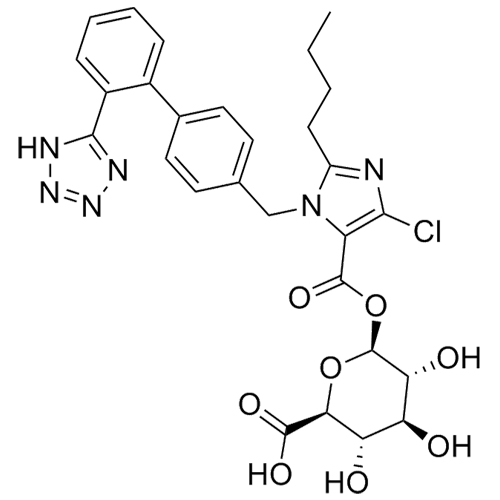
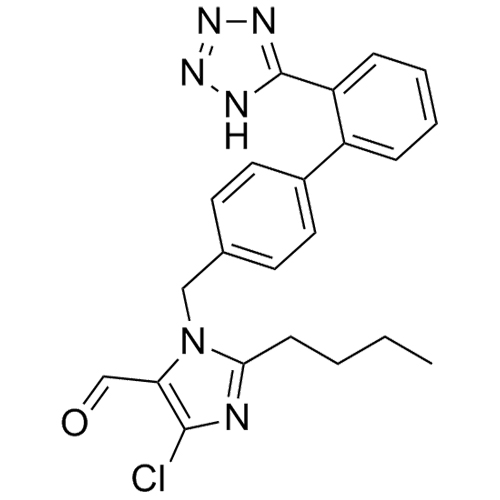
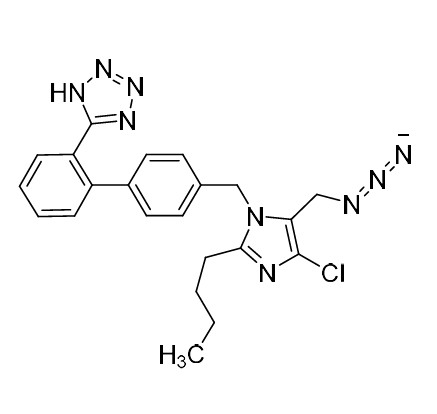
- Synonyms(1-((2'-(2H-tetrazol-5-yl)-[1,1'-biphenyl]-4-yl)methyl)-2-butyl-4-chloro-1H-imidazol-5-yl)methylacetate;2-Butyl-4-chloro-1-[[2'-(2H-tetrazol-5-yl)[1,1'-biphenyl]-4-yl]methyl]-1H-imidazole-5-methanol 5-Acetate; Losartan EP Impurity J; Losartan USP Related Compound B;O-Acetyl Losartan
- Description
(1-((2'-(2H-tetrazol-5-yl)-[1,1'-biphenyl]-4-yl)methyl)-2-butyl-4-chloro-1H-imidazol-5-yl)methylacetate;2-Butyl-4-chloro-1-[[2'-(2H-tetrazol-5-yl)[1,1'-biphenyl]-4-yl]methyl]-1H-imidazole-5-methanol 5-Acetate; Losartan EP Impurity J; Losartan USP Related Compound B;O-Acetyl Losartan
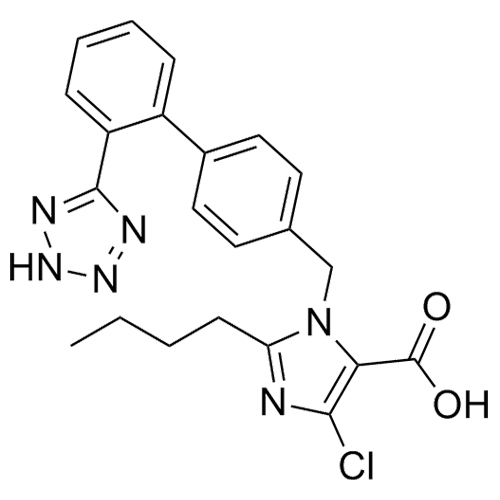
Losartan Impurity J is a fully characterized chemical compound used as a reference standard of API Losartan. The standard offered is compliant with regulatory guidelines. Losartan Impurity J is used for analytical method development, method validation (AMV), and Quality Controlled (QC) applications during synthesis and formulation stages of drug development and serves as a reference standard for traceability against pharmacopeial standards (USP or EP). Axios Research products are intended for analytical purposes only and are not for human use. CAS - 1006062-27-6


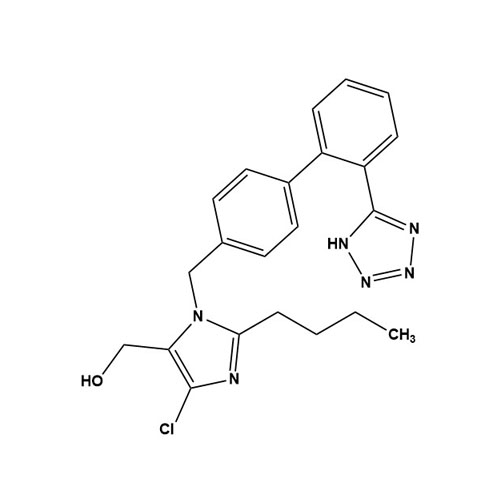
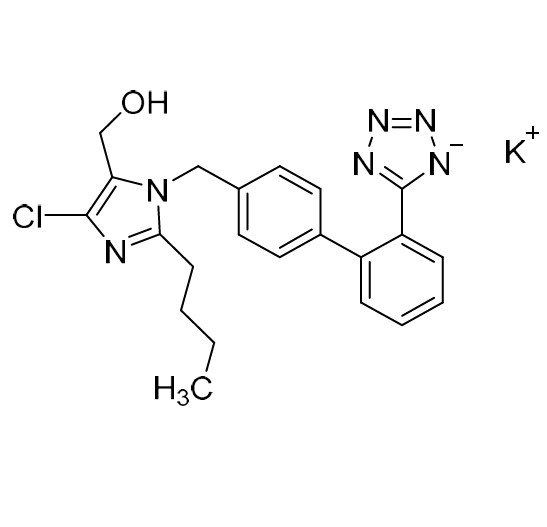
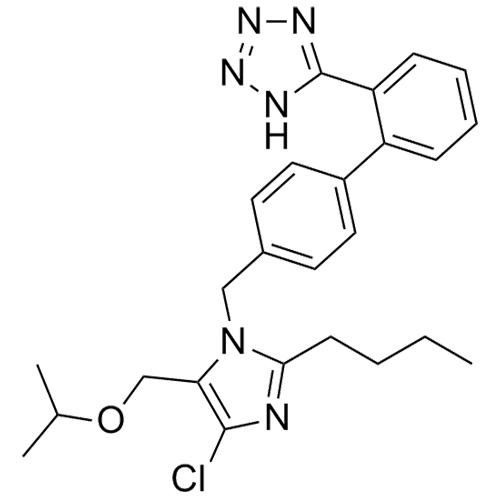
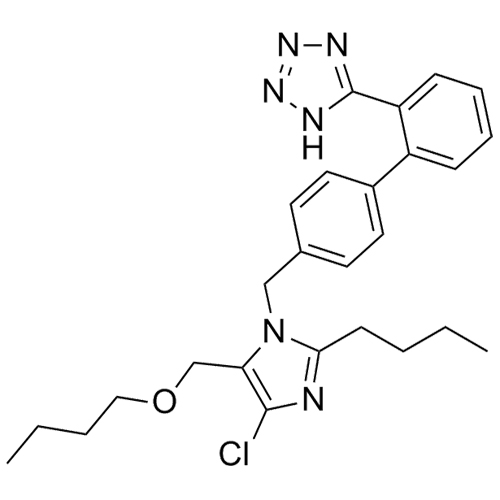
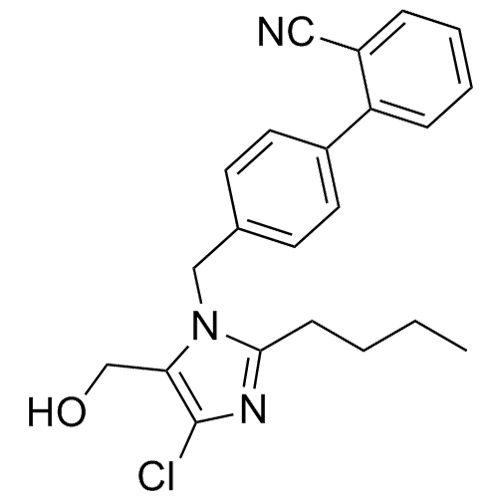
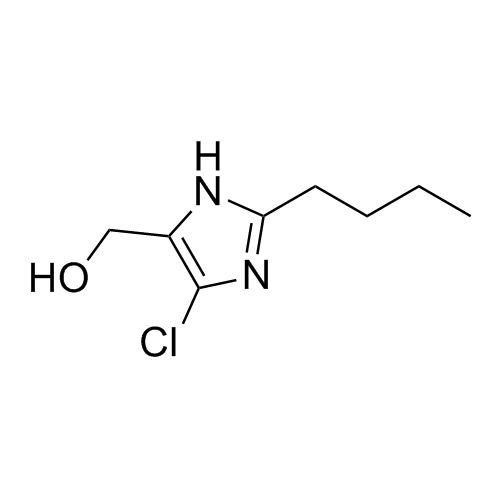
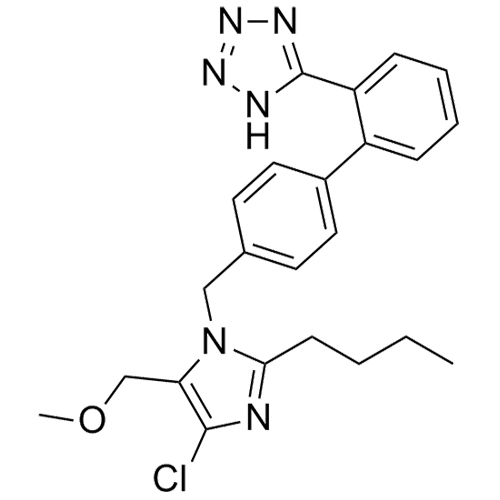
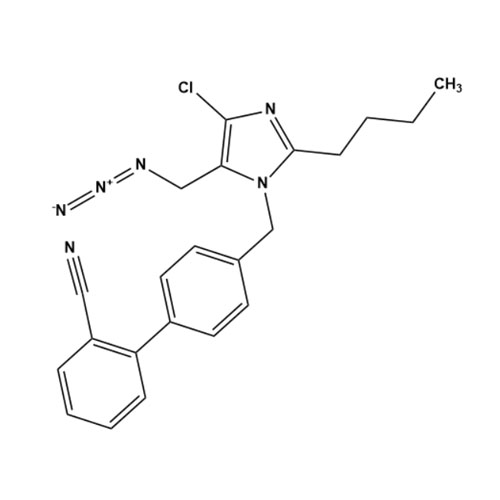
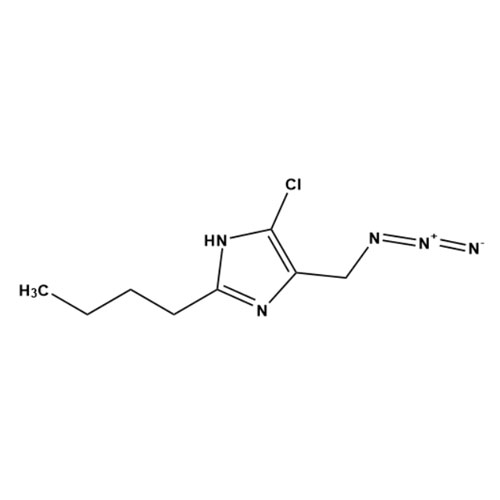
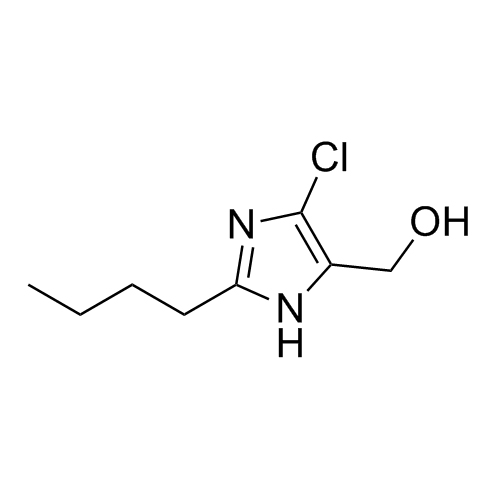
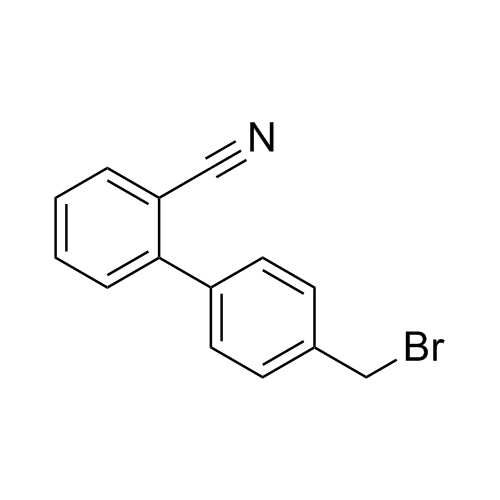
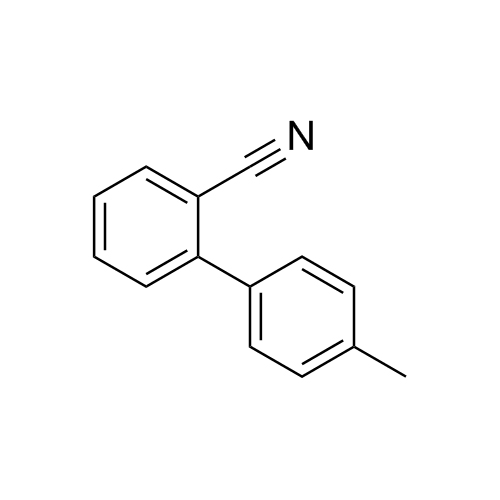
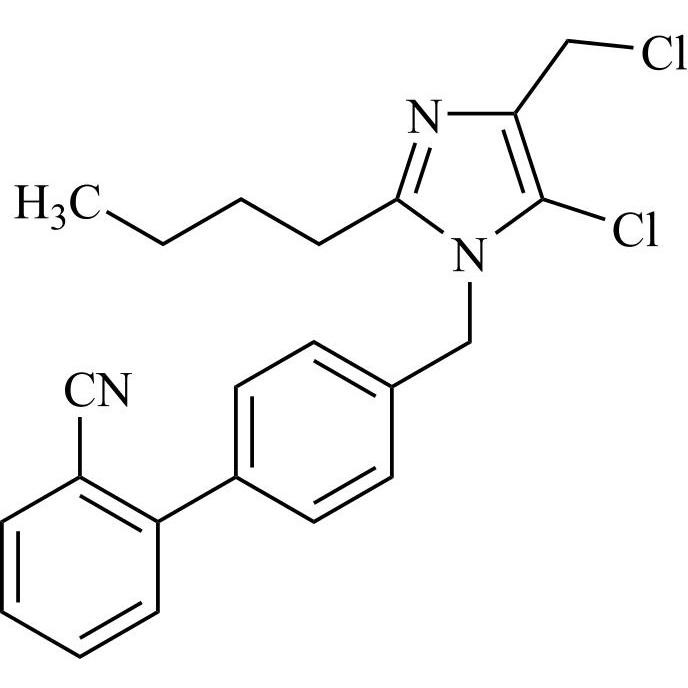
![Show details for Des[2'-(1H-tetrazol-5-yl)] 2-cyanolosartan Picture of Des[2'-(1H-tetrazol-5-yl)] 2-cyanolosartan](https://bc3c83a0874864a50753-7010fd7f2bdf81ab41ee28479491701e.ssl.cf2.rackcdn.com/media//catalog/AR-B17902.jpg?size=256)