- Synonyms(3R,5S,E)-7-(4-(4-fluorophenyl)-2-((E)-2-(4-(4-fluorophenyl)-6-isopropyl-2-(N-methylmethylsulfonamido)pyrimidin-5-yl)-N-methylvinylsulfonamido)-6-isopropylpyrimidin-5-yl)-3,5-dihydroxyhept-6-enoic acid
- Description
Related products
Rosuvastatin Impurity H Sodium Salt (Mixture of Diastereomers)
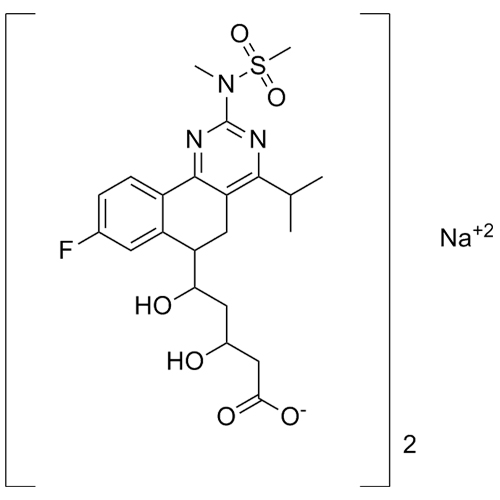
M.F.
M.W. 960 22.99
CAT# AR-R00999
CAS# NA
Trial & Method Development - Separation of Rosuvastatin Impurity H Sodium Salt Diastereomers
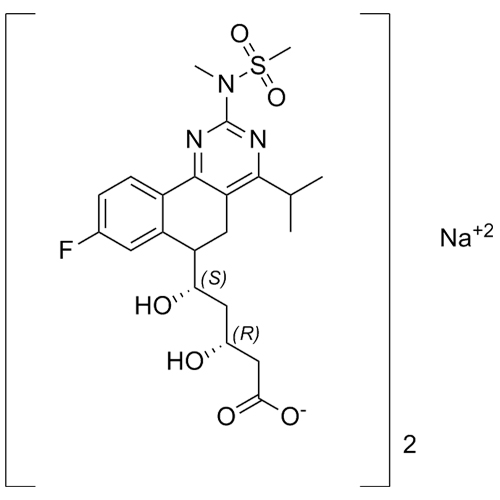
M.F. -
M.W. -
CAT# AR-R00996
CAS# NA
Rosuvastatin Impurity 6 Calcium Salt (6,7-Dihydro Rosuvastatin Calcium Salt)
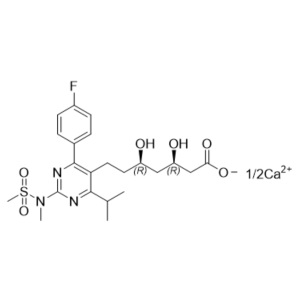
M.F.
M.W. 482.55; 40.08
CAT# AR-R10051
CAS# NA
Rosuvastatin EP Impurity C Calcium Salt
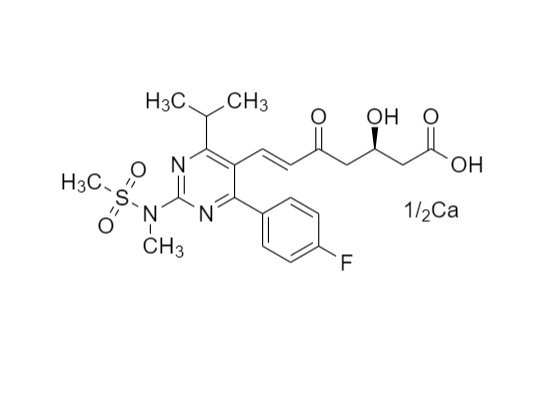
M.F.
M.W. 479.5 : 1/2(40.1)
CAT# AR-R09936
CAS# 1422514-05-3
Rosuvastatin EP Impurity E Calcium Salt
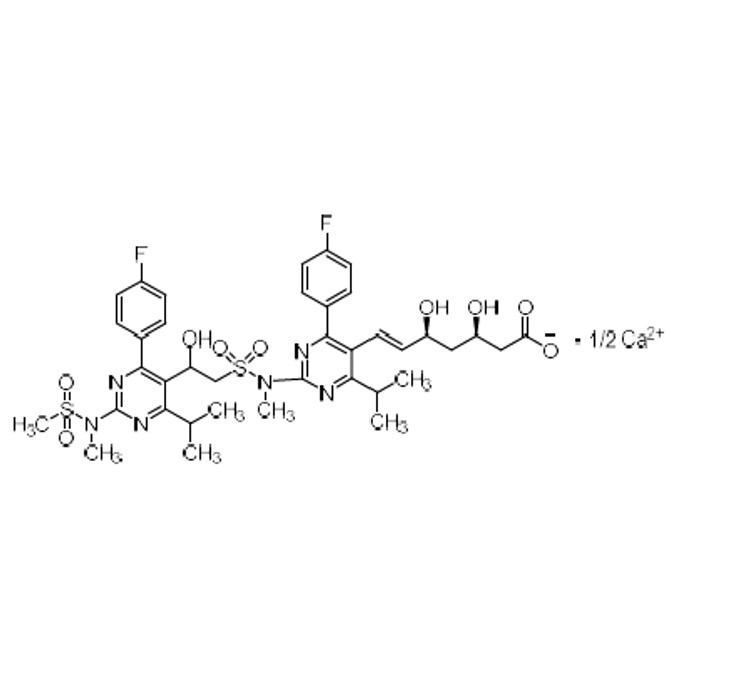
M.F.
M.W. 831.93; 1/2 (40.08)
CAT# AR-R02067
CAS# 3079028-42-2
Rosuvastatin EP Impurity B (Na Salt, Mix diastereomers)
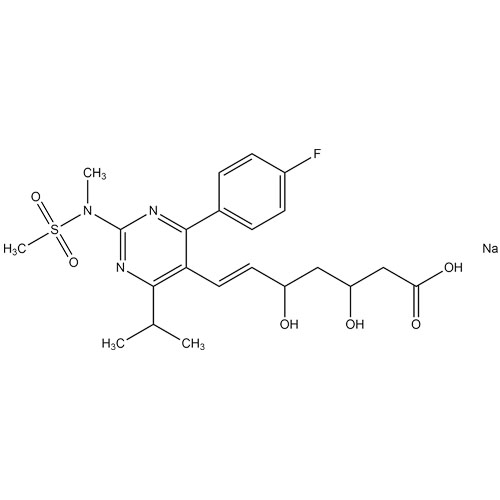
M.F.
M.W. 481.5 : 23.0
CAT# AR-R10009
CAS# 1007871-86-4
Rosuvastatin (3S,5R) Enantiomer (Calcium salt)
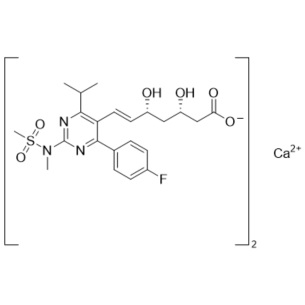
M.F.
M.W. 2(481.5); 40.08
CAT# AR-R09961
CAS# 2414245-11-5
Rosuvastatin Impurity 6 Calcium Salt (6,7-Dihydro Rosuvastatin Calcium Salt)
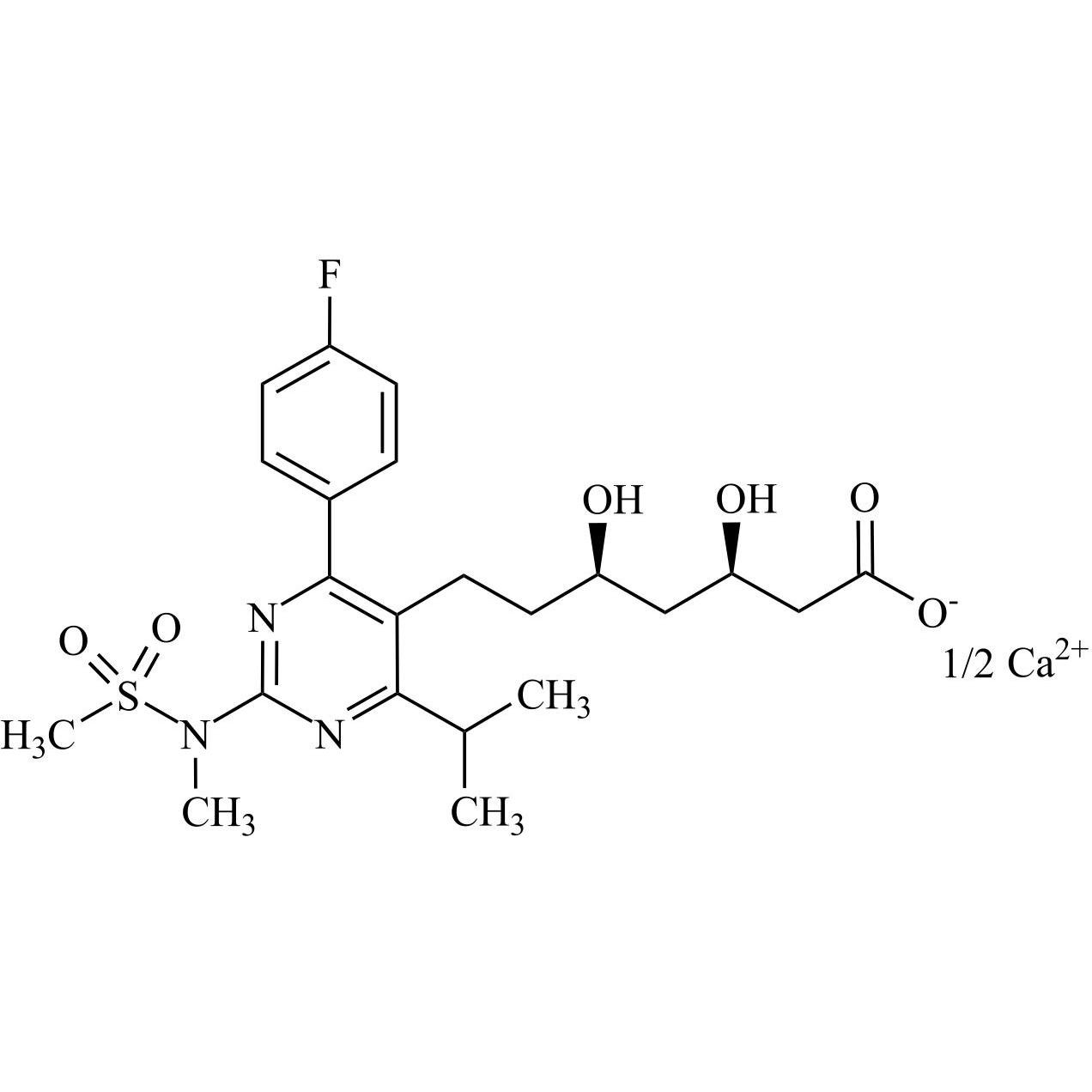
M.F.
M.W. 482.55 0.5*40.08
CAT# AR-R11526
CAS# NA
Rosuvastatin Impurity 28 Sodium Salt (Mixture of Diastereomers)
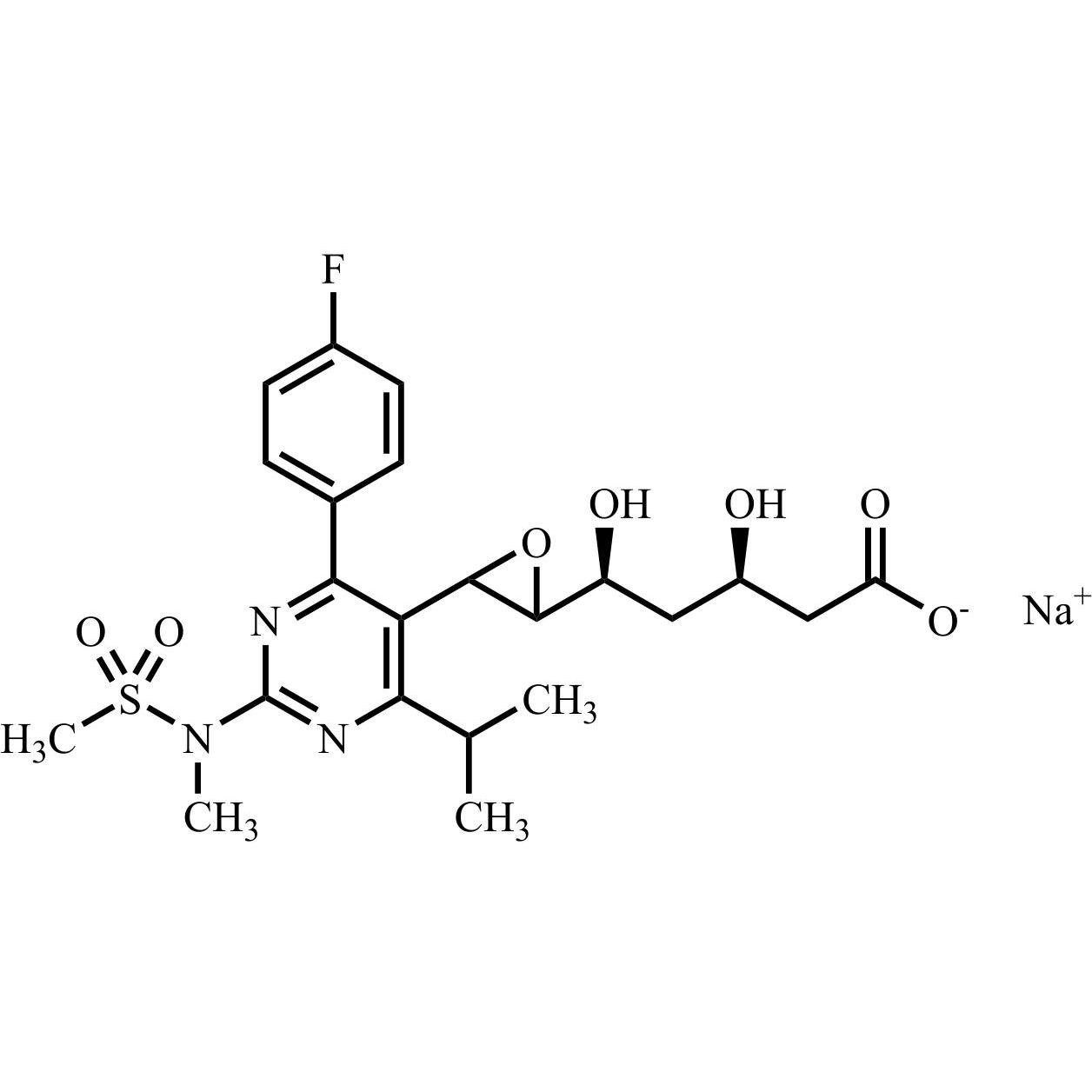
M.F.
M.W. 496.54 22.99
CAT# AR-R11533
CAS# NA
4R,6S)-6-(chloromethyl)-4-Hydroxytetrahydro-2H-Pyran-2-One
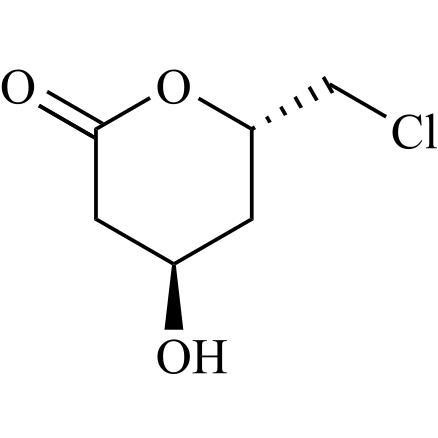
M.F.
M.W. 164.59
CAT# AR-C08599
CAS# 391218-14-7
Rosuvastatin EP Impurity L Sodium Salt (Mixture of Diastereomers)
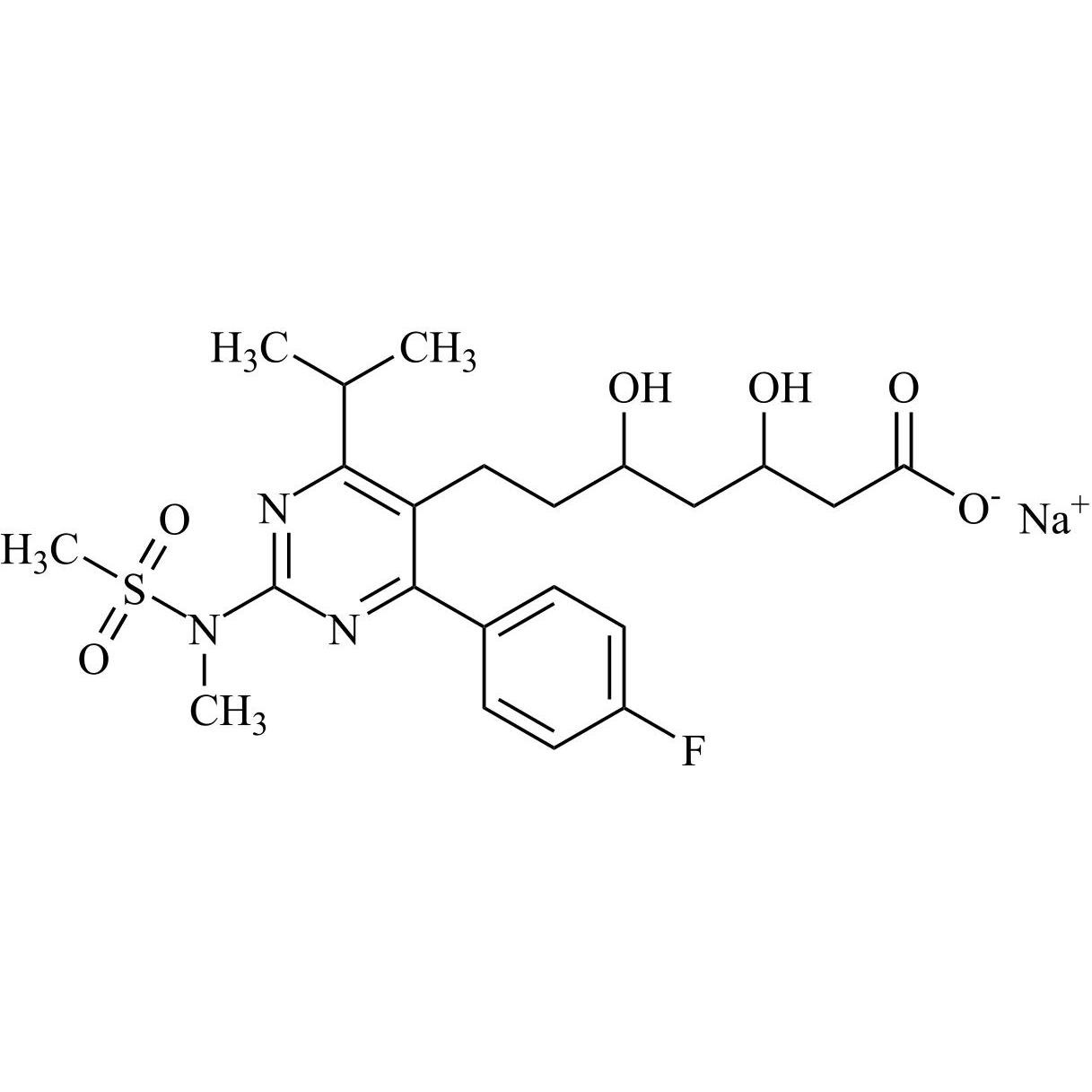
M.F.
M.W. 482.55 22.99
CAT# AR-R11509
CAS# NA
Rosuvastatin EP Impurity G Sodium Salt (ent-Rosuvastatin Sodium Salt)
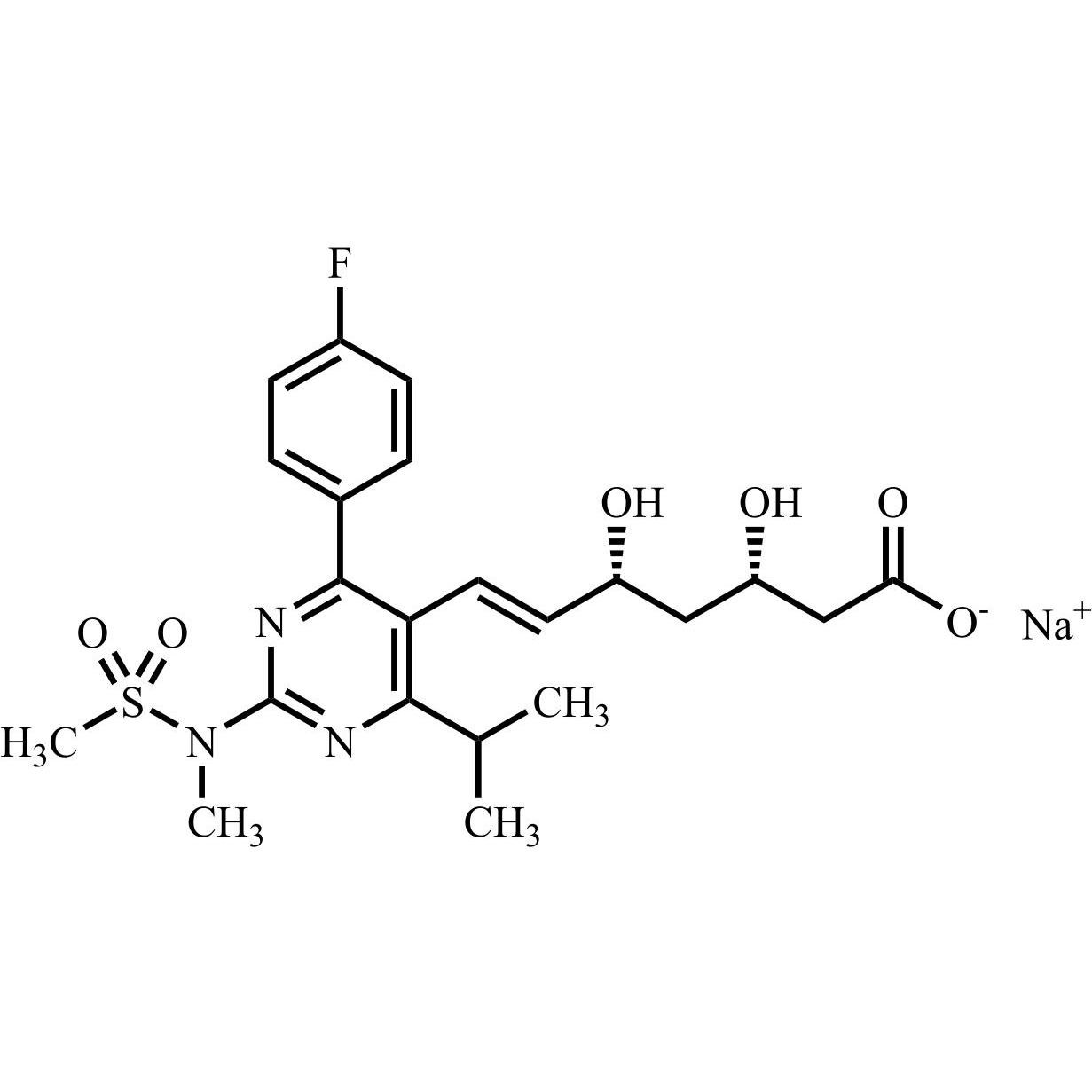
M.F.
M.W. 480.54 22.99
CAT# AR-R11511
CAS# NA
Rosuvastatin EP Impurity E Sodium Salt (Mixture of Diastereomers)
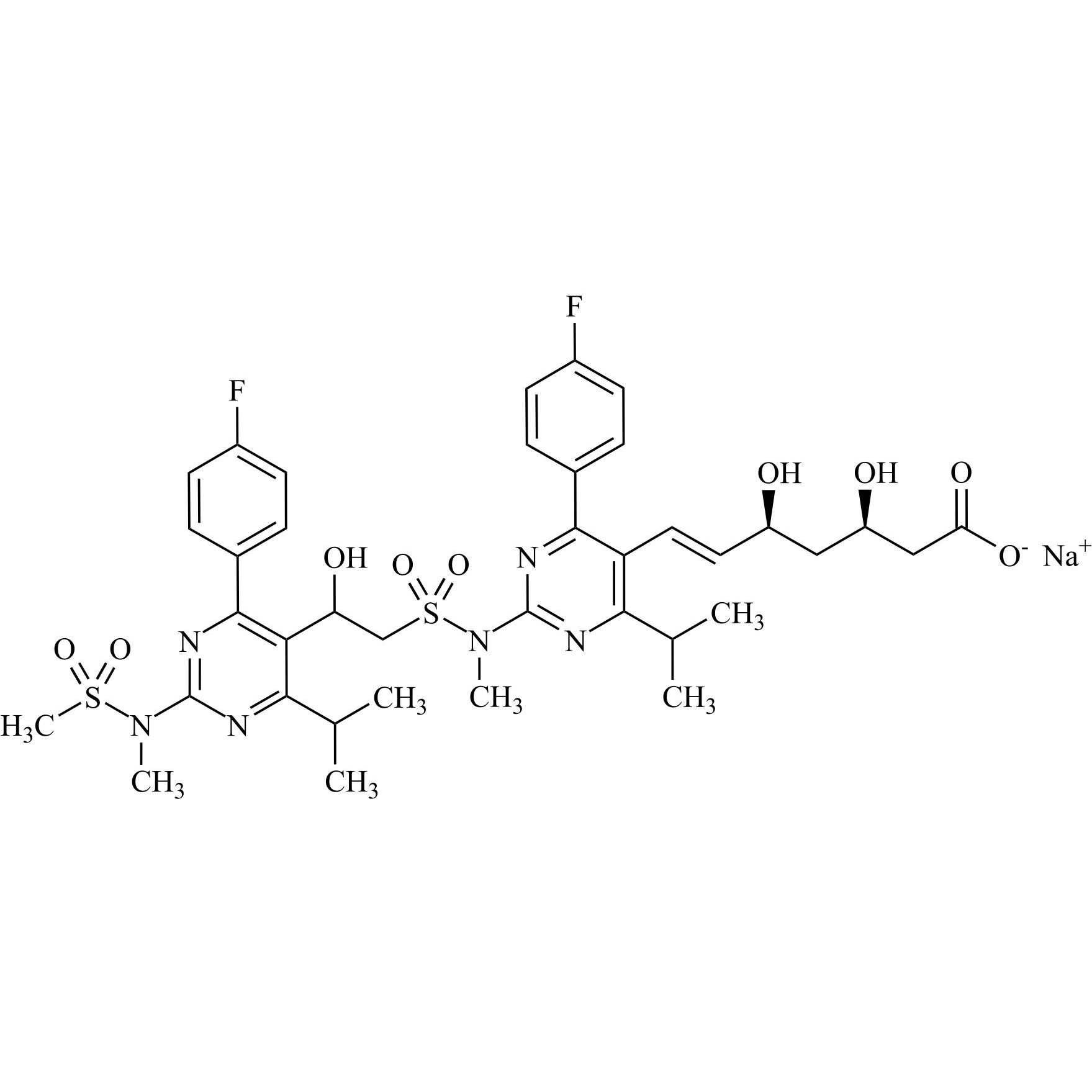
M.F.
M.W. 831.93 22.99
CAT# AR-R11516
CAS# NA
rac-Rosuvastatin EP Impurity D-d6 (rac-Rosuvastatin Lactone-d6)
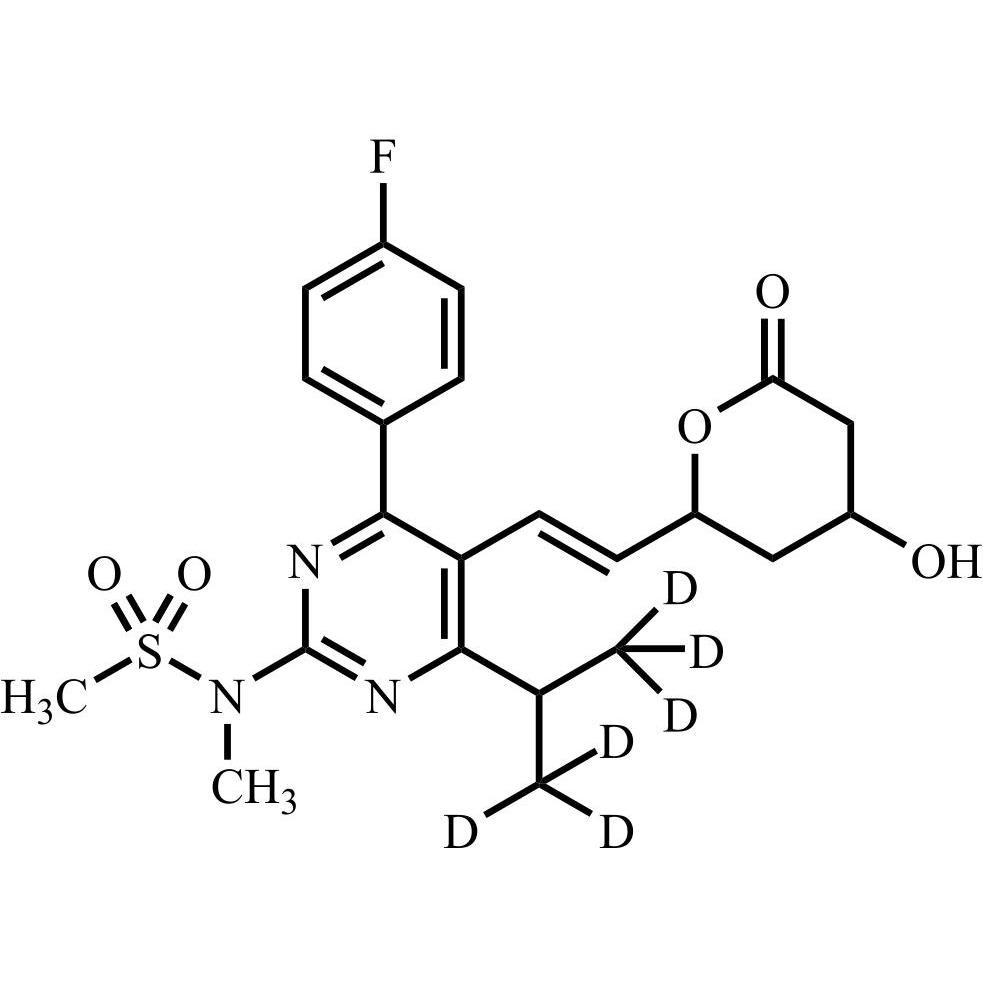
M.F.
M.W. 469.57
CAT# AR-R11443
CAS# NA